Summary :
- Alembic said it has so far received 15 product approvals this year.
- Alembic Pharma received approval from the US health regulator to market Selexipag tablets, used to treat high blood pressure, in the American market.
- Selexipag Tablets are indicated for the treatment of pulmonary arterial hypertension (PAH).
Alembic Pharmaceuticals on Wednesday said it has received approval from the US health regulator to market Selexipag tablets, used to treat high blood pressure, in the American market.
The company said it has received tentative approval from the US Food and Drug Administration (USFDA) for its abbreviated new drug application (ANDA) for Selexipag tablets in strengths of 200 mcg, 400 mcg, 600 mcg, 800 mcg, 1,400 mcg, and 1,600 mcg.

Alembic Pharma gets USFDA nod for Hypertension Drug
Alembic’s product is the generic equivalent to the reference listed drug product (RLD) Uptravi.
Selexipag Tablets are indicated for the treatment of pulmonary arterial hypertension (PAH).
According to IQVIA, Selexipag tablets have an estimated market size of USD 461 million for 12 months ending September 2021.
Selexipag
Selexipag, sold under the brand name Uptravi, is a medication developed by Actelion for the treatment of pulmonary arterial hypertension (PAH). Selexipag and its active metabolite, ACT-333679 (or MRE-269, the free carboxylic acid), are agonists of the prostacyclin receptor, which leads to vasodilation in the pulmonary circulation. It is taken by mouth or administered intravenously.
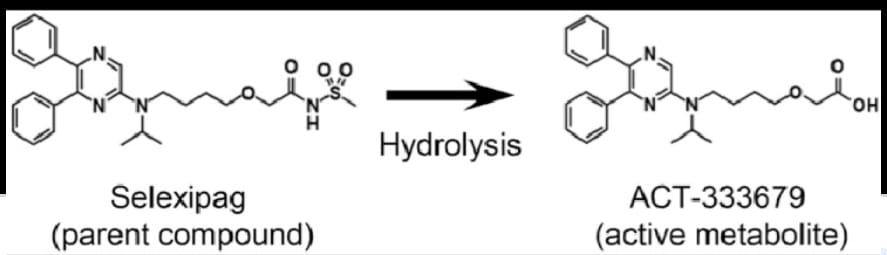
Prodrug is converted into active Compound for Action
Contraindications
In Europe, use of Selexipag together with strong inhibitors of the liver enzyme CYP2C8, such as Gemfibrozil, is contraindicated because it increases concentrations of Selexipag twofold, and its active metabolite 11-fold, potentially leading to more adverse effects.
Mechanism of action
Selexipag acts on the prostacyclin receptor of lung tissue. It is selective for the prostacyclin receptor. The binding of Selexipag to this receptor leads to three major effects: increased vasodilation of the arteries, decreased cell proliferation and inhibition of platelet aggregation, all beneficial in the treatment of pulmonary arterial hypertension.
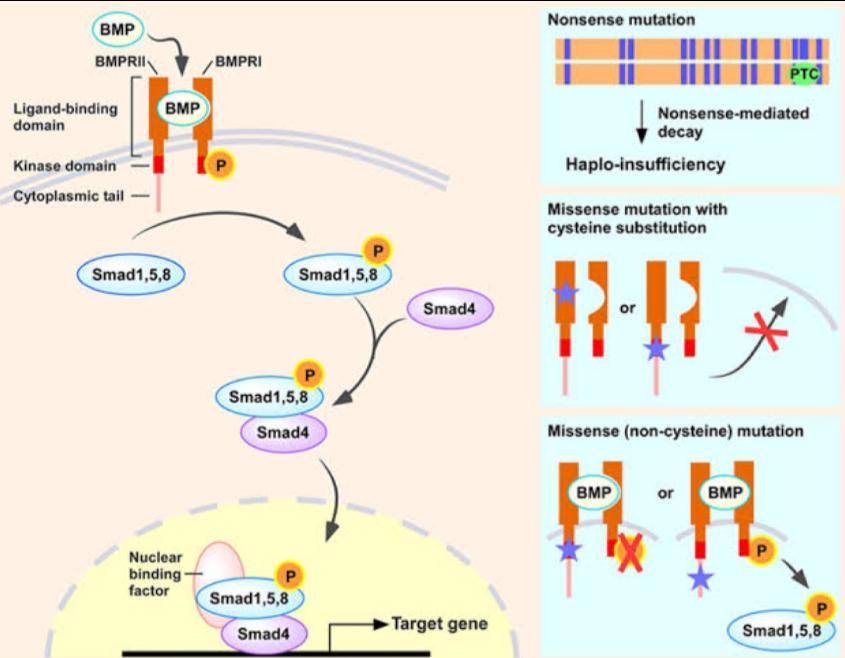
Mechanism of Action of Selexipag
Uses
Selexipag is used to treat high blood pressure in the lungs (pulmonary arterial hypertension-PAH). It is used to help slow down worsening of PAH and to decrease the chance of needing treatment in a hospital. Selexipag works by making it easier for blood to flow through the arteries in your lungs. This effect helps increase your ability to exercise.
Side Effects
Headache, nausea, vomiting, diarrhea, loss of appetite, flushing, jaw pain, joint/muscle pain, or pain in your arms or legs may occur. If any of these effects last or get worse, tell your doctor or pharmacist promptly.
Remember that this medication has been prescribed because your doctor has judged that the benefit to you is greater than the risk of side effects. Many people using this medication do not have serious side effects.
A very serious allergic reaction to this drug is rare. However, get medical help right away if you notice any symptoms of a serious allergic reaction, including: rash, itching/swelling (especially of the face/tongue/throat), severe dizziness, trouble breathing.
About Alembic Pharmaceuticals
Alembic Pharmaceuticals Ltd. is an Indian multinational pharmaceutical company headquartered in Vadodara city of Gujarat – India. Alembic Pharmaceuticals Ltd. is involved in manufacture of pharmaceutical products, pharmaceutical substances and intermediates. It is also termed to be a market leader in macrolides segment of anti-infective drugs in India.
The company has its headquarters and Corporate Office situated in Vadodara, Gujarat – India while its manufacturing facilities are located at Panelav, Karakhadi in Gujarat and Sikkim, India. Its Panelav plant houses active pharmaceutical ingredients (APIs) and formulation manufacturing, while its Sikkim plant is involved in manufacture of formulations for Indian and non-regulated export markets.
Products and services
Alembic manufactures and sells pharmaceutical and active pharmaceutical ingredients (APIs). It also offers international and domestic formulations, with branded and generic formulations being under its domestic formulations
For more Information: Sign in Websites for Agrochemical & Pharmaceutical Databases:
Website : https://www.chemrobotics.com/ (Agrochemical Databases)
Website : https://chemroboticspharma.com/ (Pharmaceutical Databases)

