Summary ;
-Oncoheroes will fund and advance the clinical development of both dovitinib and stenoparib in pediatric cancers, utilizing Allarity’s DRP® companion diagnostics
-Allarity has submitted a new drug application (NDA) for the U.S. approval of dovitinib in renal cell carcinoma (RCC) together with the Dovitinib-DRP® companion diagnostic
Allarity Therapeutics, Inc. (Nasdaq: ALLR) (“Allarity” or the “Company”), a clinical-stage biopharmaceutical company developing novel oncology therapeutics together with drug-specific DRP® companion diagnostics for personalized cancer care, and Oncoheroes Biosciences, Inc. (“Oncoheroes”), a clinical-stage biotechnology company advancing new therapies for childhood cancers, today announced that they have entered into licensing agreements under which Oncoheroes will acquire exclusive, global development rights to Allarity’s therapeutic candidates dovitinib, a pan-targeted kinase inhibitor (pan-TKI), and stenoparib, a PARP inhibitor, and assume responsibility for their further clinical development in pediatric cancers.
Under the terms of the licensing agreements, Oncoheroes acquires global, exclusive rights to fund and conduct further clinical development of both dovitinib and stenoparib in pediatric cancers. Oncoheroes will take responsibility for pediatric cancer clinical development activities for both clinical-stage therapeutics. Allarity will support Oncoheroes’ pediatric clinical trials by providing clinical-grade drug inventory at cost and by facilitating DRP® companion diagnostic screening of pediatric patients for each drug. Under the licenses, Oncoheroes will receive commercialization rights for pediatric cancers, subject to Allarity’s first buy-back option for each program, and Allarity will receive an undisclosed upfront license fee and regulatory milestones for each program. If Allarity does not re-acquire the pediatric field rights, it will further receive certain clinical/regulatory milestone payments and royalties on sales of stenoparib and dovitinib in the pediatric cancer market from Oncoheroes. Further financial terms of the licenses were not disclosed.

Allarity Therapeutics and Oncoheroes Biosciences Sign Agreements to Advance Pediatric Cancer Development of Dovitinib and Stenoparib
Steve R. Carchedi, CEO of Allarity Therapeutics, commented, “We are very pleased to partner with Oncoheroes Biosciences to advance both dovitinib and stenoparib as potential new therapeutic options for the personalized treatment of children and adolescents with cancer. Oncoheroes is a leader in advancing new therapeutics to help address historically underserved, rare childhood cancers, and an ideal partner for Allarity in the pediatric cancer market. Our out-licensing of these pediatric development programs enables Allarity to remain focused on our top priority programs in adult cancers, while at the same time leveraging Oncoheroes’ resources, capabilities, and commitment to clinically advancing dovitinib and stenoparib in childhood cancers, together with our DRP® companion diagnostics, in a true personalized cancer care approach.”
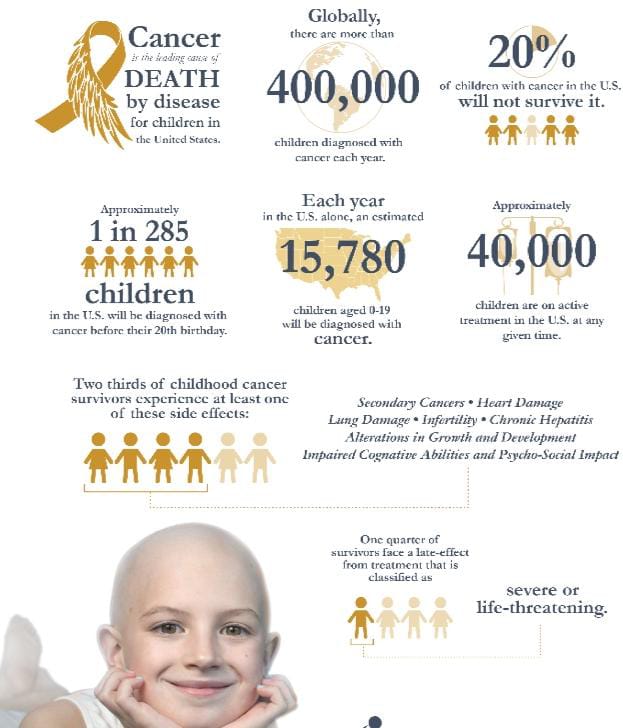
In December 2021, Allarity submitted an NDA, to the U.S. Food and Drug Administration (FDA), for marketing approval for dovitinib for the treatment of third line RCC. In April 2021, the Company submitted a premarket approval (PMA) application for use of its Dovitinib-DRP® companion diagnostic to select and treat patients most likely to respond to dovitinib. In support of its NDA filing, and in accordance with FDA requirements, the Company is also planning a clinical trial of dovitnib in pediatric patients with osteosarcoma, in partnership with Oncoheroes, where the patients will be selected with the Dovitinib-DRP® companion diagnostic. Allarity’s focus on pediatric osteosarcoma development is based on the results of two previously reported preclinical studies in which treatment with dovitinib, compared to control treatment (sucrose solution lacking dovitinib), increased the median survival time of mouse models of osteosarcoma by 50% and antitumor growth activity was observed for dovitinib as a single agent.
Ricardo Garcia, CEO & Founder of Oncoheroes Biosciences, further commented, “Oncoheroes is a mission-driven company committed to deliver more effective and safer treatments for children and adolescents with cancer. We are excited to partner with Allarity on these co-development programs. These fit perfectly with our goal of becoming the partner of choice for life sciences companies with drug candidates that have the potential to treat pediatric cancers. We are confident that this collaborative model will create powerful synergies to accelerate pediatric drug development and bring tangible benefits to younger cancer patients.”
Allarity is currently evaluating stenoparib for the treatment of advanced ovarian cancer in a Phase 2 clinical trial at the Dana-Farber Cancer Institute (Boston, MA U.S.A.), and additional U.S. and European trial sites, using the Stenoparib-DRP® companion diagnostic to guide patient enrollment and improve therapeutic outcome. In prior clinical testing of more than 60 patients, stenoparib was well tolerated with a demonstrated acceptable safety profile. Through use of DRP® patient selection, Allarity aims to provide a superior clinical benefit to ovarian cancer patients receiving stenoparib as compared to other approved PARP inhibitors. Moreover, there is increasing evidence pointing to the potential use of PARP inhibitors in the treatment of various pediatric cancers.
About Allarity Therapeutics
Allarity Therapeutics, Inc. (Nasdaq: ALLR) develops drugs for personalized treatment of cancer guided by its proprietary and highly validated companion diagnostic technology, the DRP® platform. The Company has a mature portfolio of five drug candidates, including: Stenoparib, a PARP inhibitor in Phase 2 development for ovarian cancer; Dovitinib, a pan-TKI submitted for NDA review by the FDA for the 3rd line treatment of renal cell carcinoma; IXEMPRA® (Ixabepilone), a microtubule inhibitor approved in the U.S. for the 2nd line treatment of metastatic breast cancer and in Phase 2 development, in Europe, for the treatment of the same indication; LiPlaCis®, a liposomal formulation of cisplatin in Phase 2 development for metastatic breast cancer; and 2X-111, a liposomal formulation of doxorubicin in Phase 2 development for metastatic breast cancer and/or glioblastoma multiforme (GBM). The LiPlaCis® and 2X-111 programs are partnered, via out-license, to Smerud Medical Research International AS. In 2021, Allarity sold the global rights to Irofulven, a DNA-damaging agent in Phase 2 for prostate cancer, back to Lantern Pharma, Inc. The Company maintains an R&D facility in Hoersholm, Denmark.
About the Drug Response Predictor – DRP® Companion Diagnostic
Allarity uses its drug-specific DRP® to select those patients who, by the genetic signature of their cancer, are found to have a high likelihood of responding to the specific drug. By screening patients before treatment, and only treating those patients with a sufficiently high DRP® score, the therapeutic response rate can be significantly increased. The DRP® method builds on the comparison of sensitive vs. resistant human cancer cell lines, including transcriptomic information from cell lines combined with clinical tumor biology filters and prior clinical trial outcomes. DRP® is based on messenger RNA from patient biopsies. The DRP® platform has proven its ability to provide a statistically significant prediction of the clinical outcome from drug treatment in cancer patients in 37 out of 47 clinical studies that were examined (both retrospective and prospective), including ongoing, prospective Phase 2 trials of Stenoparib and IXEMPRA®. The DRP® platform, which can be used in all cancer types and is patented for more than 70 anti-cancer drugs, has been extensively published in peer reviewed literature.
About Oncoheroes Biosciences
Oncoheroes is a ground-breaking biotech company exclusively focused on the discovery and development of better drugs for children and adolescents with cancer. Our vision is to deliver benefits to young cancer patients and create value in the process. The company is headquartered in Boston, US, with a discovery lab in Barcelona, Spain. Oncoheroes is actively looking for in-licensing opportunities in the pediatric oncology space while working to generate new proprietary assets for a number of pediatric cancer indications with high unmet medical needs.
Forward-looking Statements
Certain statements contained in this press release may constitute forward-looking statements. For example, forward-looking statements are used when discussing our expected clinical development programs and clinical trials. These forward-looking statements are based only on current expectations of management, and are subject to significant risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements, including the risks and uncertainties related to the progress, timing, cost, and results of clinical trials and product development programs; difficulties or delays in obtaining regulatory approval or patent protection for product candidates; competition from other biotechnology companies; and our ability to obtain additional funding required to conduct our research, development and commercialization activities. In addition, the following factors, among others, could cause actual results to differ materially from those described in the forward-looking statements: changes in technology and market requirements; delays or obstacles in launching our clinical trials; changes in legislation; inability to timely develop and introduce new technologies, products and applications; lack of validation of our technology as we progress further and lack of acceptance of our methods by the scientific community; inability to retain or attract key employees whose knowledge is essential to the development of our products; unforeseen scientific difficulties that may develop with our process; greater cost of final product than anticipated; loss of market share and pressure on pricing resulting from competition; and laboratory results that do not translate to equally good results in real settings, all of which could cause the actual results or performance to differ materially from those contemplated in such forward-looking statements.
For more Information: Sign in Websites for Agrochemical & Pharmaceutical Databases:
Website : https://www.chemrobotics.com/ (Agrochemical Databases)
Website : https://chemroboticspharma.com/ (Pharmaceutical Databases)

