Synopsis-
- Otezla trumped placebo in adults with moderate to severe genital psoriasis and moderate to severe plaque psoriasis, top-line results from Amgen’s phase 3 Discreet study show.
- The PDE4 inhibitor also met its secondary endpoints, charting “meaningful and significant” improvements on measures of itchiness, quality of life and more.
- Otezla is awaiting a December FDA decision to move into milder plaque psoriasis, which would open the door to an additional 1.5 million patients. The drug is currently approved in moderate to severe plaque psoriasis, plus psoriatic arthritis and Behcet’s disease.
With a potential Bristol Myers rival looming on the horizon, Amgen’s inflammatory disease med Otezla is hustling to cement its position in psoriasis. Now, the drug has notched a win in a trial for a highly stigmatized disease that can affect more than half of psoriasis patients.

Amgen ‘s Otezla wins for Psoriasis
Otezla trumped placebo in adults with moderate to severe genital psoriasis and moderate to severe plaque psoriasis, top-line results from Amgen’s phase 3 Discreet study show. The PDE4 inhibitor also met its secondary endpoints, charting “meaningful and significant” improvements on measures of itchiness, quality of life and more.
The data put Otezla in the running for another psoriasis nod and the drug could certainly use the boost. Otezla, which Amgen bought from Celgene in 2019 for $13.4 billion, may soon face pressure from Bristol Myers Squibb’s TYK2 inhibitor deucravacitinib. BMS expects to launch its rival in the second half of 2022.
Meanwhile, Otezla is awaiting a December FDA decision to move into milder plaque psoriasis, which would open the door to an additional 1.5 million patients. The drug is currently approved in moderate to severe plaque psoriasis, plus psoriatic arthritis and Behcet’s disease.
Genital psoriasis is a highly stigmatizing disease that can affect up to 63% of psoriasis patients, David Reese, M.D., executive vice president of research and development at Amgen, said in a release. Treatment often comes in the form of topical therapies, but many patients still find it tough to manage their condition, which has prompted experts to call for systemic treatments, he said.
About 125 million people worldwide, or 2% to 3% of the total population, have psoriasis, the National Psoriasis Foundation says. That means there could be around 75 million people worldwide suffering from genital psoriasis.
Amgen’s Discreet study used the modified static Physician’s Global Assessment of Genitalia (sPGA-G) which tracks the severity of genital psoriasis for its primary endpoint. Success was defined as an sPGA-G response score of 0, meaning clear, or 1, meaning almost clear, with at least a 2-point reduction from baseline at week 16.
Patients who received 30 mg Otezla twice a day hit clinically meaningful and statistically significant improvements over placebo on their sPGA-G responses, Amgen said. The drug also met all its secondary endpoints, leading to “meaningful and significant” improvements on measures of genital psoriasis itch, affected body surface area, quality of life and more.
About Genital Psoriasis
Genital psoriasis is a type of the skin condition psoriasis that you get on or around your genitals. Sometimes that may be the only place you have it. But many people have itchy, red patches on other parts of their body at the same time.
Genital psoriasis isn’t an STD, and it’s not catching. But it can change the way you feel about your body and put a damper on your love life. It’s also more uncomfortable and harder to treat than other types of psoriasis.
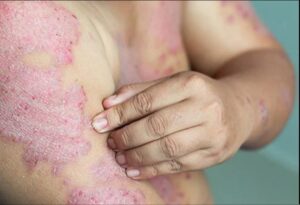
Skin condition Psoriasis
Cause :
Some of the most common are:
- Infections
- Skin injuries
- Being overweight or obese
- Smoking
- Heavy drinking
- Stress
- Certain medicines
- Cold weather
Symptoms :
Psoriasis in areas like
- Pubic area
- Upper thighs
- Creases between your thighs and groin
- Anus and crease between buttocks
About Otezla

Apremilast, sold under the brand name Otezla among others, is a medication for the treatment of certain types of psoriasis and psoriatic arthritis. It may also be useful for other immune system-related inflammatory diseases. The drug acts as a selective inhibitor of the enzyme phosphodiesterase 4 (PDE4) and inhibits spontaneous production of TNF-alpha from human rheumatoid synovial cells. It is taken by mouth.
Apremilast is indicated in the United States for the treatment of adults with active psoriatic arthritis, people with moderate to severe plaque psoriasis who are candidates for phototherapy or systemic therapy, and adults with oral ulcers associated with Behçet’s disease.
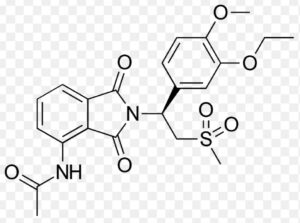
Structure of Apremilast
In the European Union, Apremilast alone or in combination with disease-modifying Antirheumatic drugs (DMARDs), is indicated for the treatment of active psoriatic arthritis (PsA) in adults who have had an inadequate response or who have been intolerant to a prior DMARD therapy. It is also indicated for the treatment of moderate to severe chronic plaque psoriasis in adults who failed to respond to, have a contraindication to, or are intolerant of other systemic therapies, including cyclosporine, methotrexate, or psoralen and ultraviolet-A light.
About Amgen
Amgen Inc. (formerly Applied Molecular Genetics Inc.) is an American multinational biopharmaceutical company headquartered in Thousand Oaks, California.One of the world’s largest independent biotechnology companies, Amgen was established in Thousand Oaks, California, in 1980. Amgen’s Thousand Oaks staff in 2017 numbered 5,125 (7.5% of total city employment) and included hundreds of scientists, making Amgen the largest employer in Ventura County. Focused on molecular biology and biochemistry, its goal is to provide a healthcare business based on recombinant DNA technology

Logo of Amgen Inc.
In 2018, the company’s largest selling product lines were Neulasta, an immunostimulator used to prevent infections in patients undergoing cancer chemotherapy and Enbrel, a tumor necrosis factor blocker used in the treatment of rheumatoid arthritis and other autoimmune diseases. Other products include Epogen, Aranesp, Sensipar/Mimpara, Nplate, Vectibix, Prolia and XGEVA. Amgen sponsors the Tour of California.


