Highlight
Shionogi & Co., Ltd. (Head Office: Osaka, Japan; President and CEO: Isao Teshirogi, Ph.D.; hereafter “Shionogi”) today announced that Shionogi has completed the analysis of primary endpointsa), b) in the Phase 2b part of a Phase 2/3 clinical trial of S-217622, an orally administered antiviral drug for COVID-19, and has filed for manufacture and sales approval, requesting review under the conditional approval system in Japan.
a) Change from baseline on Day 4 (after the 3rd dose) in SARS-CoV-2 viral titer
b) Time-weighted average change in the total score of 12 COVID-19 symptoms from the initiation of
administration (Day 1) up to 120 hours (Day 6)
Oral antiviral medications, in addition to vaccines, are expected to play an important role in treating coronavirus disease 2019 (COVID-19), which is caused by infection with the severe acute respiratory disease coronavirus-2 (SARS-CoV-2).
Novel Covid Drug
These drugs must have significant antiviral activity, as well as target specificity, oral bioavailability, and metabolic stability. Although several antiviral compounds have been reported as possible SARS-CoV-2 inhibitors in vitro, only a few of these drugs have been shown to be effective in vivo.
Ensitrelvir (code name S-217622, brand name Xocova), is a new inhibitor of the SARS-CoV-2 major protease (Mpro), also known as 3C-like protease, has been shown to reduce the viral load and help alleviate the severity of SARS-CoV-2 in infected hamsters. In cells, low nanomolar to sub-micromolar doses of S-217622 suppress viral growth. In hamsters, oral treatment of S-217622 showed excellent pharmacokinetic qualities and hastened recovery from acute SARS-CoV-2 infection.

Covid Drug Xocova
S-217622 also demonstrated antiviral effectiveness against SARS-CoV-2 variants of concern (VOCs), such as the highly pathogenic Delta variant and the newly discovered Omicron variant. Overall, these findings show that S-217622, which is an antiviral drug that is currently being tested in Phase II/III clinical trials, has impressive antiviral efficiency and effectiveness against SARS-CoV-2 and could be a viable oral treatment option for COVID-19.
Ensitrelvir
Ensitrelvir (code name S-217622, brand name Xocova) is an antiviral drug developed by Shionogi in partnership with Hokkaido University, which acts as an orally active 3C-like protease inhibitor for the treatment of COVID-19 infection. It is taken by mouth, and has been successfully tested against the recently emerged Omicron variant

Xocova Covid Drug of Japan
Quick Notch : Exclusive ChemRobotics Data
Name – Ensitrelvir
Brand Name –Xocova
Category – Antiviral Drug
Code Name – S-217622
Originator – drug developed by Shionogi in partnership with Hokkaido University (Japan )
CAS No – 2647530-73-0
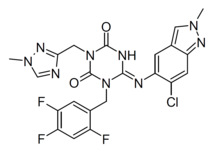
IUPAC Name – 6-[(6-chloro-2-methylindazol-5-yl)amino]-3-[(1-methyl-1,2,4-triazol-3-yl)methyl]-1-[(2,4,5-trifluorophenyl)methyl]-1,3,5-triazine-2,4-dione
Structure –
Ensitrelvir Structure
Synonyms : BDBM513874bioRxiv20220126.477782, S-217622
Formula – C26H21ClF3N9O6
Mol. Wt – 647.95
Route of Administration – Oral
Route of Synthesis :
Route of Synthesis of Ensitrelvir(S-217622, S 217622, Xocova, Shionogi Covid-19 pill)
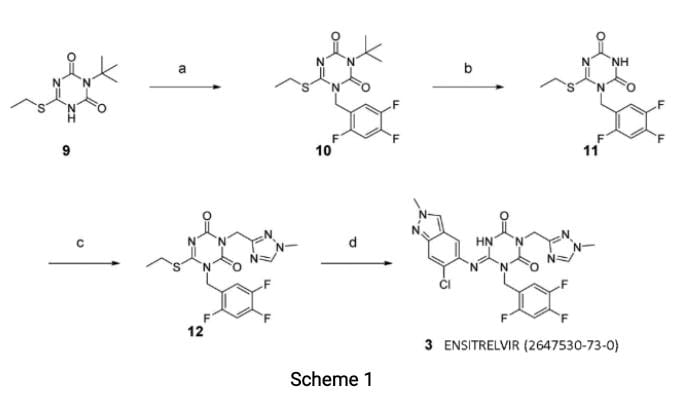
Ensitrelvir (S-217622, S 217622, Xocova, Shionogi Covid-19 pill) was synthesized in the literature as described in Scheme 1. Starting from known compound9,an alkylation with 1-(bromomethyl)-2,4,5-trifluorobenzene gave 10 in 93% yield. Then,
the 3-tert-Bu group was removed and the triazole unit was introduced, and the substitutionof the SEt moiety with the indazole unit finally gave Ensitrelvir (S-217622).
For Full Route Visit : https://chemroboticspharma.com/pharmVetPat
Clinical Data
- A Phase 1 study of S-217622 in healthy adult participants (jRCT2031210202) Japan Registry of Clinical Trials Web Site 2021, July 16
- Phase 2/3 clinical trial is a randomized, placebo-controlled, double-blind study in 428 SARS-CoV-2 infected subjects with mild/moderate symptoms (419 in Japan and 9 in South Korea)
Phase 2a Results :
- A more rapid decline in viral titer and viral RNA.
- Phase 2b Results
- A more rapid decline in viral titer and viral RNA.
- On day 4 of treatment, less than 10% of patients had a positive viral titer.
- There was a trend toward improvement in COVID-19 symptoms, according to the time-weighted average change in the total score of 12 COVID-19 symptoms between days 1–6 from the start of treatment, although no statistically significant difference with placebo was noted, but such a difference was registered for respiratory symptoms (stuffy or runny nose, sore throat, cough, shortness of breath).
Clinical Data of Antiviral
- This study was conducted mainly in infected patients after the Omicron variant wave of the epidemic, and its main purpose is to confirm the antiviral effect and clinical symptom improvement of S-217622 (2 dose groups) when orally administered once daily for 5 days.
- Shionogi Presents Phase 2/3 Clinical Trial Results of the COVID-19 Therapeutic Drug S-217622
Approval – It became the first Japanese domestic pill to treat COVID-19, third to be regulatorally approved in Japan; in February 2022
Antiviral effect
・At both doses, the S-217622 arms showed a significant difference on day 4 (after the 3rd dose), in comparison to the placebo arm, with respect to each of the following:
→Rapid reduction in viral titer (achieved primary endpoint)
→The proportion of subjects with positive viral titer, which was below 10% in the S-217622 treated arms, and decreased to a greater degree than seen in the Phase 2a part, 1 in comparison with the placebo arm
Clinical symptom improvement
・No significant difference in the time-weighted average change in the total score of 12 COVID-19 symptoms from initiation of administration up to 120 hours while they changed in the direction of improvement (primary endpoint not achieved)
・Of the 12 symptoms, a significant improvement effect in the total score of respiratory symptoms (stuffy or runny nose, sore throat, cough, shortness of breath), highly characteristic symptoms in the population enrolled in this study, was observed at both doses
Safety
・TEAE and treatment-related TEAE were consistent with those observed in the Phase 2a part, and no new adverse events of concern were observed at the time of this analysis
Mechanism of Action-
Xocova (ensitrelvir) is an oral antiviral drug for the treatment of COVID-19 infection caused by the new coronavirus SARS-CoV-2.
The active ingredient in Xocova is ensitrelvir (S-217622), a slow-molecule non-peptidomimetic non-covalent orthosteric selective inhibitor of 3C-like protease (3CLpro), also known as main protease (Mpro), an enzyme necessary for coronavirus replication.
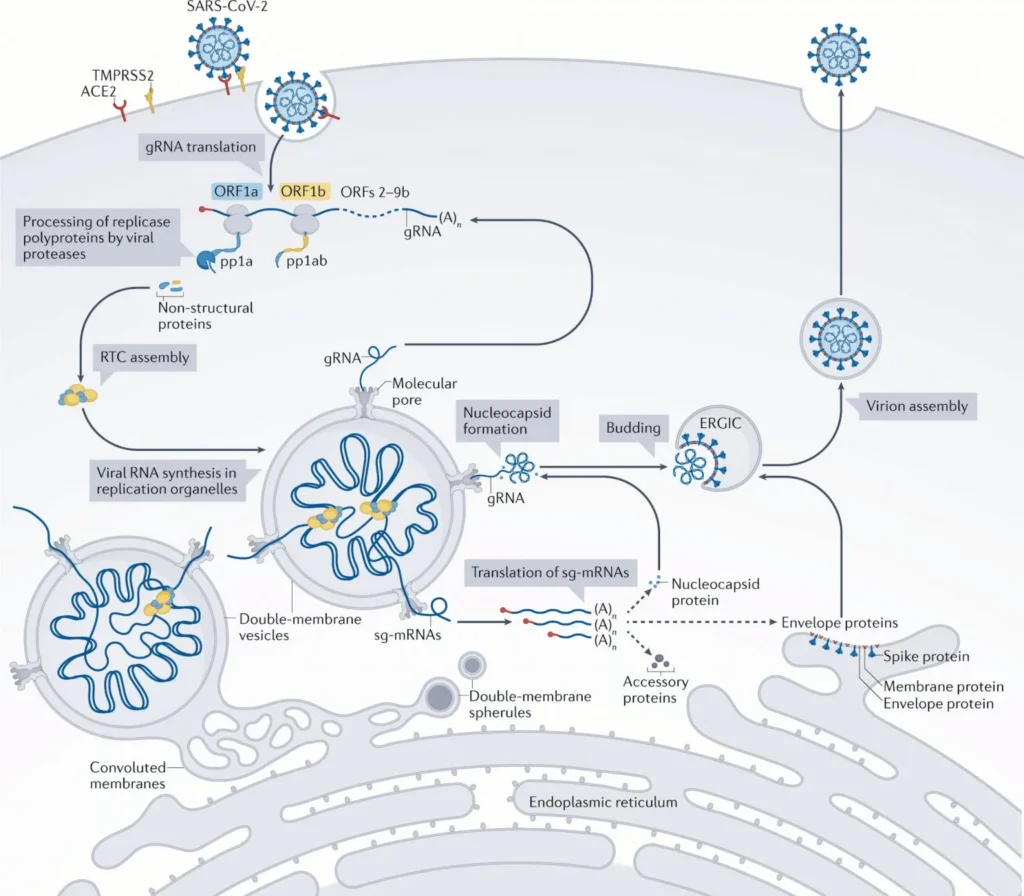
Ensitrelvir Mechanism
SARS-CoV-2, being an enveloped positive-sense (+) RNA virus, enters host cells by binding its S protein to angiotensin-converting enzyme 2 (ACE2) on the cells and then releasing genomic RNA (gRNA) into the cytoplasm after uncoating. The cell’s ribosomes then translate the gRNA into the two major polyproteins, pp1a and pp1ab. Proteases embedded in viral nonstructural proteins 3 (Nsp3) and 5 (Nsp5), papain-like protease (PLpro) and chymotrypsin-like protease (3CLpro), respectively, cleave pp1a and pp1ab into 16 nonstructural proteins that assemble into replication–transcription complexes (RTCs). The latter produce new gRNAs and a set of subgenomic RNAs (sgRNAs) including open reading frames (ORFs) that encode structural viral proteins such as S protein, membrane (M), envelope (E), and nucleocapsid (N) proteins, as well as a number of accessory proteins. The newly created gRNAs are translated to produce additional nonstructural proteins, serve as a template for further RNA synthesis, or are packaged into new virions.
It is 3C-like protease inhibitor for the treatment of COVID-19 infection. It is taken by mouth, and has been successfully tested against the recently emerged Omicron variant
The nsp5 gene, which encodes Mpro, a target for S-217622, is largely conserved between each of these VOCs and VOIs, unlike the S gene, which encodes the viral spike protein. As a result of changes in the viral spike protein, S-217622 had antiviral effects against all VOCs, whereas neutralizing antibodies displayed varied reactivities to different VOCs. In Vero-TMPRSS2 cells infected with the SARS-CoV-2 Omicron variant, S-217622 retained exceptional antiviral effectiveness.
S-217622 exhibited antiviral activity in hamsters infected with various VOCs including the Alpha, Gamma, and Omicron variants. In hamsters, SARS-CoV-2 infection resulted in severe pneumonia and triggered various inflammatory responses; however, S-217622 treatment reduced inflammatory cytokine expression levels
About S-217622
S-217622, a therapeutic drug for COVID-19, is a 3CL protease inhibitor created through joint research between Hokkaido University and Shionogi. SARS-CoV-2 has an enzyme called 3CL protease, which is essential for the replication of the virus. S-217622 suppresses the replication of SARS-CoV-2 by selectively inhibiting 3CL protease. Shionogi has already been submitting the non-clinical, manufacturing/CMC data, and clinical trial data obtained so far to the PMDA. Currently the Phase 3 part of a Phase 2/3 clinical trial in patients with mild/moderate symptoms and the Phase 2b/3 part in patients with asymptomatic/only mild symptoms are in progress.
Synthesis Scheme –
- Starting from the pyrazole derivative 4, cyclization with Ethyl isocyanatoacetate and CDI was conducted, giving 5 in 90% yield.
- Then, an alkylation with 5-bromomethyl-1,2,3-trifluorobenzene followed by introduction of a 4-difluoromethoxy-2-methylaniline unit, to give 7 (40% in 2 steps).
- The ester group in 7 was hydrolyzed and then amidated with methylamine, yielding 1 (58% in 2 steps).
- Compound 2 was synthesized similarly as shown above
- S-217622 (3) was synthesized as described in Scheme 3. Starting from known compound 9,21 an alkylation with 1-(bromomethyl)-2,4,5-trifluorobenzene gave 10 in 93% yield.
- Then, the 3-tert-Bu group was removed and the triazole unit was introduced, and the substitution of the SEt moiety with the indazole unit finally gave S-217622
Implications
The viral spike protein, which develops amino acid changes across SARS-CoV-2 variants, is the target of current vaccines and monoclonal antibody treatments. Comparatively, viral Mpro is less diverse, thereby demonstrating that the susceptibility of SARS-CoV-2 VOCs to S-217622 is likely comparable.
Taken together, the SARS-CoV-2 Mpro is a promising target for antiviral therapies. Furthermore, the findings from the current study demonstrate that S-217622 has a unique potential in treating COVID-19.
About Shionogi
Shionogi & Company, Limited is a Japanese pharmaceutical company best known for developing Crestor. Medical supply and brand name also uses Shionogi. Shionogi has business roots that date back to 1878, and was incorporated in 1919.
In Japan it is particularly known as a producer of antimicrobial and antibiotics. Because of antibiotic resistance and slow growth of the antibiotic market, it has teamed up with US based Schering-Plough (merged in 2009 with Merck & Co) to become a sole marketing agent for its products in Japan.
Shionogi had supported the initial formation of Ranbaxy Pharmaceuticals, a generic manufacturer based in India. In 2012 the company became a partial owner of ViiV Healthcare, a pharmaceutical company specialising in the development of therapies for HIV.
The company is listed on the Tokyo Stock Exchange and Osaka Securities Exchange and is constituent of the Nikkei 225 stock index
References
- Press release on February 7, 2022
- “Shionogi presents positive Ph II/III results for COVID-19 antiviral S-217622”. thepharmaletter.com. 31 January 2022.
- ^ Shionogi’s new COVID pill appears to ease omicron symptoms. Nikkei Asia, 21 December 2021
- ^ Japan to consider early approval for Shionogi COVID-19 pill. Japan Times, 8 February 2022
- ^ “Japan’s Shionogi seeks approval for COVID-19 pill”. Reuters. 25 February 2022.
- ^ “Japan’s Shionogi seeks approval for COVID-19 pill”. Reuters. Reuters. 25 February 2022.


