Key points:
- India’s Everest Organics starts manufacturing Molnupiravir Active Pharmaceutical ingredient (API) for Merck’s COVID-19 pill.
- Everything you need to know about Merck`s COVID-19 pill
- The Merck pill, which could become the first oral antiviral COVID-19 treatment, forces the SARS-CoV-2 coronavirus to mutate itself to death.
- Merck said it asked the FDA to authorize emergency use of its experimental antiviral pill to treat mild to moderate Covid-19 in adults.
- Molnupiravir is a prodrug of the synthetic nucleoside derivative N4-hydroxycytidine and exerts its antiviral action through the introduction of copying errors during viral RNA replication.
- Optimus interim phase 3 data for Molnupiravir shows 78.3% efficacy.
- WHO says Merck’s antiviral Covid pill is ‘certainly good news as it awaits data.
- At the Interim Analysis, 7.3 Percent of Patients Who Received Molnupiravir Were Hospitalized Through Day 29, Compared With 14.1 Percent of Placebo-Treated Patients Who were Hospitalized or Died.
- Merck Plans to Seek Emergency Use Authorization in the U.S. as Soon as Possible and to Submit Applications to Regulatory Agencies Worldwide.
- Phase three clinical trial data showed that the drug – known as Molnupiravir– reduced the chances that patients newly diagnosed with Covid would be hospitalized by about 50%.
- The experimental drug could be available to Americans by late this year.
- a potentially game-changing advancement in the fight against the virus, which is killing an average of more than millions of people.
- “We’re looking forward to receiving the data from them,” WHO’s Maria Van Kerkhove said.

India’s Everest Organics starts making ingredients for Merck’s COVID-19 pill.
Indian generic drug maker Everest Organics started manufacturing active pharmaceutical ingredients for Merck’s oral COVID-19 treatment. News of the production rollout comes in tandem with Merck filing for emergency use authorization from the FDA for the antiviral, Molnupiravir, which would be the first newly developed oral treatment to stave off COVID-19.
“After the successful development and commercialization of various COVID-19 drugs, Everest Organics is on its path of expansion of this portfolio and hence has further developed an additional COVID-19 treatment (Molnupiravir) API in that segment,” Srikakarlapudi Sirisha, M.D., Everest’s chief executive, said in a filing (PDF) with the BSE.
The company said the production of Molnupiravir is being done at a lab scale. Merck filed for a EUA with the FDA on Monday, just 10 days after it and partner Ridgeback Biotherapeutics released data from their phase 3 trial.
Pharmaceutical major Merck and Ridgeback Biotherapeutics announced via a press release on October 1 the early results from Phase-3 trials that its anti-viral drug Molnupiravir halved the chances of hospitalization in COVID-19 patients with mild or moderate disease.
Placebo trials involve testing a drug on thousands of people, in which some of them get the drug and some — who are in a placebo group — do not. In the placebo arm, 53 patients of 14% were either hospitalized or had died, whereas in the group that got the drug, 28 — or 7.3% — were hospitalized or succumbed to the infection. After 29 days of monitoring, no deaths were reported in patients who received Molnupiravir, as compared to eight deaths in those who received placebo.
Merck’s announcement that its experimental antiviral pill is effective against the most severe outcomes of Covid-19 is “certainly good news,” a World Health Organization official said Tuesday, as the international agency awaits clinical trial data on the drug.
“We’re looking forward to receiving the data from them,” Maria Van Kerkhove, the WHO’s technical lead for Covid, said during a virtual Q&A. “I think everybody wants earlier treatment so that we prevent people from actually, you know, getting to that severe state and actually dying from the disease.”
The U.S. drugmaker said Friday that the medication – known as Molnupiravir – was shown in a phase three trial to reduce the risk of hospitalization or death by around 50% for adults with mild to moderate cases of Covid. It works by inhibiting the replication of the virus inside the body
“The extraordinary impact of this pandemic demands that we move with unprecedented urgency, and that is what our teams have done by submitting this application for Molnupiravir to the FDA within 10 days of receiving the data,” Merck CEO Robert Davis said in a press release.
Merck, which developed the drug with Ridgeback Biotherapeutics, said it is actively working with regulatory agencies worldwide to submit applications for emergency use or authorization “in the coming months.”
While vaccinations remain the best form of protection against the virus, U.S. officials and health experts hope a pill like Merck’s will keep the disease from progressing in those who do get infected and prevent trips to the hospital.
Pills like Merck’s are considered a sort of “holy grail” for treatments, Dr. Mike Ryan, executive director of the World Health Organization’s Health Emergencies Program, said at a news conference last week
Indian Everest Organic started making API for Merck’s COVID-19 pill.
India’s Everest Organics Ltd said on Tuesday it had started making the active pharmaceutical ingredient (API) for a generic version of Merck & Co’s experimental antiviral drug Molnupiravir to treat mild to moderate COVID-19.
Shares of Everest Organics jumped as much as 11.6% after the news came in, and were last up 9.9% at 330 rupees in a weak Mumbai market. The Indian bulk drugs manufacturer joins Divi’s Laboratories Ltd as an API maker for Merck’s experimental oral drug. Merck has separately entered into voluntary licensing agreements with at least eight Indian generic drugmakers for Molnupiravir, with an aim to turn the country into a manufacturing hub for the drug.
“After the successful development and commercialization of various COVID-19 drugs such as Oseltamivir, Remdesivir… Everest Organics is on its path of expansion of this portfolio,” Chief Executive Officer Srikakarlapudi Sirisha said in a statement.
What is Molnupiravir?
Molnupiravir is a broad-spectrum antiviral that is an orally bioavailable prodrug of the nucleoside analogue β-D-N4-hydroxycytidine (NHC), and exerts its antiviral action through the introduction of copying errors during viral RNA replication.
Molnupiravir was invented at Drug Innovations at Emory (DRIVE), LLC, a not-for-profit biotechnology company wholly owned by Emory University.
Molnupiravir (MK-4482/EIDD-2801) is an investigational, orally administered form of a potent ribonucleoside analog that inhibits the replication of SARS-CoV-2, the causative agent of COVID-19. Molnupiravir has been shown to be active in several preclinical models of SARS-CoV-2, including for prophylaxis, treatment, and prevention of transmission. Additionally, pre-clinical and clinical data have shown Molnupiravir to be active against the most common SARS-CoV-2 variants. Molnupiravir was invented at Drug Innovations at Emory (DRIVE), LLC, a not-for-profit biotechnology company wholly owned by Emory University, and is being developed by Merck & Co., Inc. in collaboration with Ridgeback Biotherapeutics. Ridgeback received an upfront payment from Merck and also is eligible to receive contingent payments dependent upon the achievement of certain developmental and regulatory approval milestones. Any profits from the collaboration will be split between the partners equally. Since licensed by Ridgeback, all funds used for the development of Molnupiravir have been provided by Merck and by Wayne and Wendy Holman of Ridgeback.
Molnupiravir is also being evaluated for post-exposure prophylaxis in MOVe-AHEAD, a global, multicentre, randomized, double-blind, placebo-controlled Phase 3 study, which is evaluating the efficacy and safety of Molnupiravir in preventing the spread of COVID-19 within households
Mechanism of Action (MOA):
Molnupiravir is metabolized into a ribonucleoside analog that resembles cytidine, β-D-N 4-Hydroxycytidine 5′-triphosphate (also called EIDD-1931 5′-triphosphate). During replication, the virus’s RNA polymerase enzyme incorporates EIDD-1931 5′-triphosphate into newly-made RNA instead of using real cytidine.
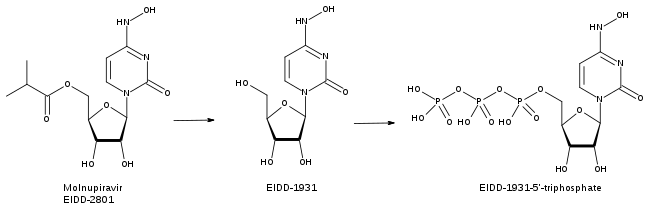
Molnupiravir can swap between two forms (tautomers), one of which mimics cytidine (C) and the other of which mimics uridine (U). When the viral RNA polymerase attempts to copy RNA containing Molnupiravir, it sometimes interprets it as C and sometimes as U. This causes a massive number of mutations in all downstream viral copies that exceed the threshold the virus can survive, an effect called viral error catastrophe or lethal mutagenesis.
Top, a G.C base pair with three hydrogen bonds. Bottom, an A.U base pair with two hydrogen bonds. Molnupiravir can mimic both C and U. The wiggly lines stand for the connection to the pentose sugar and point in the direction of the minor groove.
Molnupiravir’s active metabolite EIDD-1931 has been found to produce DNA mutations as well in a mammalian cell culture assay, raising concerns for potential carcinogenic and teratogenic effects.
How to use Molnupiravir?
Molnupiravir will be able to be taken orally by patients at home after they receive a prescription from their doctor. A course of treatment will be eight 200 milligram capsules a day for five days; four capsules in the morning and four capsules in the afternoon. A patient can choose whether they want to take the medicine with or without food, as it doesn’t appear to affect the medicine in the body.
It’s unclear whether a positive COVID test will be required before a prescription can be issued. This is something that will be decided by the TGA.
How all this started.
Molnupiravir began as a possible therapy for the Venezuelan equine encephalitis virus at Emory University’s non-profit company DRIVE (Drug Innovation Ventures at Emory) in Atlanta. But in 2015, DRIVE’s chief executive George Painter offered it to a collaborator, virologist Mark Denison at Vanderbilt University in Nashville, Tennessee, to test against coronaviruses. “I was pretty blown away by it,” Denison remembers. He found that it worked against multiple coronaviruses: MERS and mouse hepatitis virus2.
Painter also recruited his collaborator Plemper to test the drug against influenza and respiratory syncytial virus. After the pandemic hit, however, plans changed. DRIVE licensed the compound to Ridgeback Biotherapeutics in Miami, Florida. Plemper, too, pivoted to coronaviruses and tested the compound in ferrets. It silenced the virus’s ability to replicate, he says, but it also suppressed the virus’s transmission from infected ferrets to uninfected ones3. Merck’s data hint that might also be true in humans: Molnupiravir appeared to shorten the duration of SARS-CoV-2’s infectivity in trial participants with the virus.
The importance of Molnupiravir by Mercks.
Merck (NYSE: MRK), known as MSD outside the United States and Canada, and Ridgeback Biotherapeutics today announced that Molnupiravir (MK-4482, EIDD-2801), an investigational oral antiviral medicine, significantly reduced the risk of hospitalization or death at a planned interim analysis of Phase 3 MOVe-OUT trial in at-risk, non-hospitalized adult patients with mild-to-moderate COVID-19. At the interim analysis, Molnupiravir reduced the risk of hospitalization or death by approximately 50%; 7.3% of patients who received Molnupiravir were either hospitalized or died through Day 29 following randomization (28/385), compared with 14.1% of placebo-treated patients (53/377); p=0.0012. Through Day 29, no deaths were reported in patients who received Molnupiravir, as compared to 8 deaths in patients who received placebo. At the recommendation of an independent Data Monitoring Committee and in consultation with the U.S. Food and Drug Administration (FDA), recruitment into the study is being stopped early due to these positive results. Merck plans to submit an application for Emergency Use Authorization (EUA) to the U.S. FDA as soon as possible based on these findings and plans to submit marketing applications to other regulatory bodies worldwide.
“More tools and treatments are urgently needed to fight the COVID-19 pandemic, which has become a leading cause of death and continues to profoundly affect patients, families, and societies and strain health care systems all around the world. With these compelling results, we are optimistic that Molnupiravir can become an important medicine as part of the global effort to fight the pandemic and will add to Merck’s unique legacy of bringing forward breakthroughs in infectious diseases when they are needed most. Consistent with Merck’s unwavering commitment to save and improve lives, we will continue to work with regulatory agencies on our applications and do everything we can to bring Molnupiravir to patients as quickly as possible,” said Robert M. Davis, chief executive officer and president, Merck. “On behalf of all of us at Merck, I thank our network of clinical investigators and patients for their essential contributions to the development of Molnupiravir”
“With the virus continuing to circulate widely, and because therapeutic options currently available are infused and/or require access to a healthcare facility, antiviral treatments that can be taken at home to keep people with COVID-19 out of the hospital are critically needed,” said Wendy Holman, chief executive officer of Ridgeback Biotherapeutics. “We are very encouraged by the results from the interim analysis and hope Molnupiravir if authorized for use, can make a profound impact in controlling the pandemic. Our partnership with Merck is critical to ensuring rapid global access if this medicine is approved, and we appreciate the collaborative effort to reach this important stage of development.”
Would it be accessible to the world?
An effective oral antiviral would be an incredible asset in the fight against COVID-19, but it’s not yet clear whether Molnupiravir will be accessible to all. “Are we going to be in a situation where the price is reasonable in low- and middle-income countries?” asks Rachel Cohen, the North American executive director at the Drugs for Neglected Diseases initiative.
The United States has agreed to purchase 1.7 million courses of Molnupiravir for US$1.2 billion, which works out to about $700 per 5-day course. That’s far less than the price of Remdesivir or monoclonal antibodies, but still too costly for much of the world. Merck, which is co-developing the compound with Ridgeback, has struck licensing agreements with five Indian manufacturers of generic drugs. Those deals allow the manufacturers to set their own price in India and 100 other low- and lower-middle-income countries.
Merck and Co earlier this year announced that it signed non-exclusive voluntary licensing agreements for Molnupiravir with five generic manufacturers in India. It was done to make sure that the availability of the drug in more than 100 low- and middle-income countries could be accelerated following approvals from their regulatory agencies.
Malaysia health minister Khairy Jamaluddin said that negotiations between Merck and the Malaysian government have begun to procure the new drugs. The US government has also contracted to buy 1.7 million doses of Merck’s medication.
But even if poorer countries can afford the drug, they might not have the diagnostic capacity to use it properly. If Molnupiravir needs to be given in the first five days after symptom onset, “that requires that we are able to actually rapidly diagnose people”, Cohen says. For many developing countries — and even some wealthy ones — “that is actually a huge challenge”.
Drug Structure
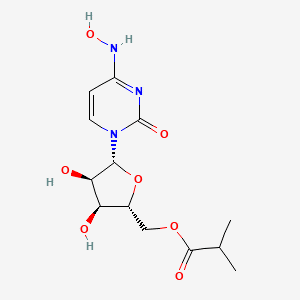
Molnupiravir
- MOLECULAR FORMULA: C13H19N3O7
- MOLECULAR WEIGHT: 329.31 g/mol
- IUPAC: [(2R, 3S, 4R, 5R)-3,4-dihydroxy-5-[4-(hydroxyamino)-2-oxopyrimidin-1-yl]oxolan-2-yl]methyl 2-methylpropanoate
- CAS RN. 2349386-89-4 (Deleted CAS RN); 2492423-29-5 (Molnupiravir)
IP Scenario: Patent Number: WO2019113462A1 Assignee: Emory University, United States Publication Date: 2019-06-13 Est.Exp.: 2038-12-07 Application Number: WO2018-US64503 Application Date: 2018-12-07 Equivalents: WO2019113462; CA3082191; AU2018378832; IL274155; KR2020071119; CN111372592; GB2581936; EP3706762; BR112020010581; JP6804790; GB2581936; JP2021505530; KR2248165; KR2021050594; AU2018378832; AU2018378832; GB2590198; US20200276219; IN202017019418; JP2021042235 Indian Equivalents: IN202017019418 Title: N4-hydroxycytidine and derivatives and anti-viral uses related thereto Abstract: This disclosure relates to certain N4-hydroxycytidine derivatives, pharmaceutical compositions, and methods related thereto. In certain embodiments, the disclosure relates to the treatment or prophylaxis of viral infections, such as Eastern, Western, and Venezuelan Equine Encephalitis (EEE, WEE and VEE, respectively), Chikungunya fever (CHIK), Ebola, Influenza, RSV, and Zika virus infection with the disclosed compounds.
Clinical Trial Data:
The Phase III portion of an ongoing Phase II/III trial of Merck and Ridgeback Biotherapeutics’ molnupiravir in mild-to-moderate Covid-19 has enrolled around 1,100 to 1,200 patients of its 1,550-patient target, an investigator said. The trial could complete enrolment in one to two months, assuming worldwide infection continues to spread at its current rate.
A Merck spokesperson said the trial’s enrolment is on track; November is the company’s best guess for when the trial will have enough events for final analysis. The placebo-controlled Phase III MOVe-OUT study has a primary endpoint of the percentage of patients who are hospitalized and/or die over 29 days.
The trial is enrolling adults with laboratory-confirmed, mild-to-moderate Covid-19, and at least one risk factor associated with poor disease outcomes. Participants are randomized to either 800mg molnupiravir or placebo twice daily for five days. ClinicalTrials.gov lists the trial’s estimated primary completion date as 8 November.
Hyderabad Optimus pharma.

Optimus Pharma`s results of clinical trial
Hyderabad-based Optimus Pharma’s interim results of phase III clinical trials for orally administered Molnupiravir capsules on patients with mild Covid-19 patients showed reduced viral load and 78.3 percent efficacy. The company said it has submitted the data to the Indian drugs regulator Drugs Controller General of India (DCGI) on Sunday.
Views of Indian Pharmaceuticals on Molnupiravir.
Two drugmakers in the country have requested permission to end their late-stage trials on Merck & Co’s experimental antiviral drug Molnupiravir in moderate COVID-19 patients.
The two drugmakers, Aurobindo Pharma Ltd and MSN Laboratories plan to continue late-stage trials of the drug for those with mild COVID-19, the drug regulator’s expert committee said on Friday.
The two companies separately sought permission to end trials in the case of moderate COVID-19 patients after having submitted interim clinical trial data around the effectiveness of the drug in treating this category of patients, the committee disclosed, throwing into question the efficacy of the experimental drug in improving outcomes for patients with moderate cases of COVID-19. Separately, a source at the drug regulator told Reuters that Molnupiravir had shown no “significant efficacy” against moderate COVID-19 cases
Shares in Merck soared last week after it and partner Ridgeback Biotherapeutics said an interim analysis of a late-stage clinical trial on Molnupiravir showed the medicine nearly halved the risk of hospitalization or death for patients with mild or moderate COVID-19. It was not immediately clear whether the drugmakers and Merck used identical criteria to define moderate COVID-19 cases.
Aurobindo Pharma, MSN, and Merck did not immediately reply to Reuters’ requests for comment.
Merck has entered into voluntary licensing agreements with at least eight Indian drugmakers for Molnupiravir, with an aim to turn the country into a manufacturing hub for the drug to supply low- and middle-income nations.
Aurobindo Pharma has been conducting a clinical trial on the drug in 100 patients with moderate COVID-19 since August this year according to its trial details, moderate patients included those with fever, coughing, breathing difficulties, and oxygen deficiency.
Of the eight Indian firms, five – Dr. Reddy’s Laboratories, Cipla, Sun Pharma, Torrent Pharmaceuticals, and Emcure Pharmaceuticals – are conducting a joint trial for the antiviral drug only in mild COVID-19 patients in an outpatient setting. The other licensed company, Hetero, in early July announced interim data from its own late-stage trial in treating mild COVID-19 patients and submitted an application for emergency use approval for the same.
Hetero is separately conducting a study on moderate COVID-19 patients and it has said clinical results on the same will be shared in due course.
Other Antivirals are in the works.
Gilead Sciences is developing a pill version of Remdesivir. And Denison suspects that if the antiviral were given to people as early as Molnupiravir is — when symptoms have only just appeared and viral loads are high — it would be similarly effective. In a study presented at IDWeek, a virtual meeting of infectious disease specialists and epidemiologists held earlier this month, researchers reported results of administering infusions of Remdesivir to people in the early stages of COVID-19 every day for three days. The number of participants in the study was small, but Remdesivir appeared to reduce hospitalizations by 87% in people at high risk of developing COVID-19.
Biotech firm Atea Pharmaceuticals in Boston, Massachusetts, also has an antiviral in the works. It was testing a nucleoside analog against hepatitis C in a clinical study when SARS-CoV-2 emerged. The pandemic paused the trial, so Atea decided to switch its focus to COVID-19. Now it has partnered with Roche in Basel, Switzerland, to develop its compound.
Pfizer, based in New York City, had a bit of a head start too. The company had been developing antivirals against SARS since the early 2000s but shelved them when the outbreak ebbed. When the COVID-19 pandemic began, “they just blew the dust off”, Luly says. Researchers are currently testing a pill form of a compound that has a mechanism of action similar to those original versions. It is in phase 2/3 trials for treating people who are newly infected.
For more information – Sign in Websites for Agrochemical & Pharmaceutical Databases:
- Website: https://www.chemrobotics.com/ (Agrochemical Databases)
- Website: https://chemroboticspharma.com/ (Pharmaceutical Databases)


