SUMMARY:
- Iptacopan has been awarded the Innovation Passport for the treatment of C3 glomerulopathy (C3G) and is currently being investigated in a number of complement-driven renal diseases and one hematological disorder for patients with high unmet need.
- The Innovation Passport is a designation for innovative medicines. It is the entry point to the Innovative Licensing and Access Pathway (ILAP), which aims to support the development of these medicines and bring them to patients more rapidly.
- Novartis is committed to ensuring their medicines reach patients who need them most and is excited to explore the opportunities through this new pathway to bring Iptacopan to eligible patients.

Novartis Pharmaceuticals UK awarded an Innovation Passport for Investigational Oral Therapy Iptacopan (LNP023).
Novartis have announced that the investigational oral therapy, Iptacopan (LNP023), has been awarded an Innovation Passport for the treatment of C3 glomerulopathy (C3G) by the MHRA, NICE, and the SMC. It is currently being investigated in a number of complement-driven renal diseases(CDRDs), alongside one haematological disorder, for patients with high unmet need.
The Innovation Passport is a designation for innovative medicines. It is the entry point to the Innovative Licensing and Access Pathway (ILAP), a new pathway aiming to accelerate the time taken to market, allowing a more rapid patient access to medicines. A single Innovation Passport can cover multiple future indications for the same active substance.
Iptacopan is an investigational, first-in-class, orally administered factor B inhibitor for the alternative pathway of the complement system. It has the potential to become the first targeted therapy to delay progression to dialysis in C3G, and was discovered at the Novartis Institute for BioMedical research. Novartis has a 35-year history in kidney transplantation treatments, however, iptacopan is the first treatment in the nephrology pipeline addressing CDRDs.
C3 glomerulopathy is a group of related conditions that cause the kidneys to malfunction. After kidney transplantation, the risk of recurrence in the transplanted kidney is over 70%, with more than a 50% chance of graft loss.
About Innovative Licensing and Access Pathway (ILAP):
ILAP was launched in January 2021 as a new pathway aiming to accelerate time to the market, facilitating patient access to medicine. It opens to commercial and non-commercial developers of medicines and provides opportunities for enhanced input from regulatory and other stakeholders. ILAP provides a single integrated platform for collaborative working between the MHRA, healthcare partners including NICE and SMC, and the medicine developer. The Innovation Passport acts as the entry point to the pathway.
What is C3 glomerulopathy?
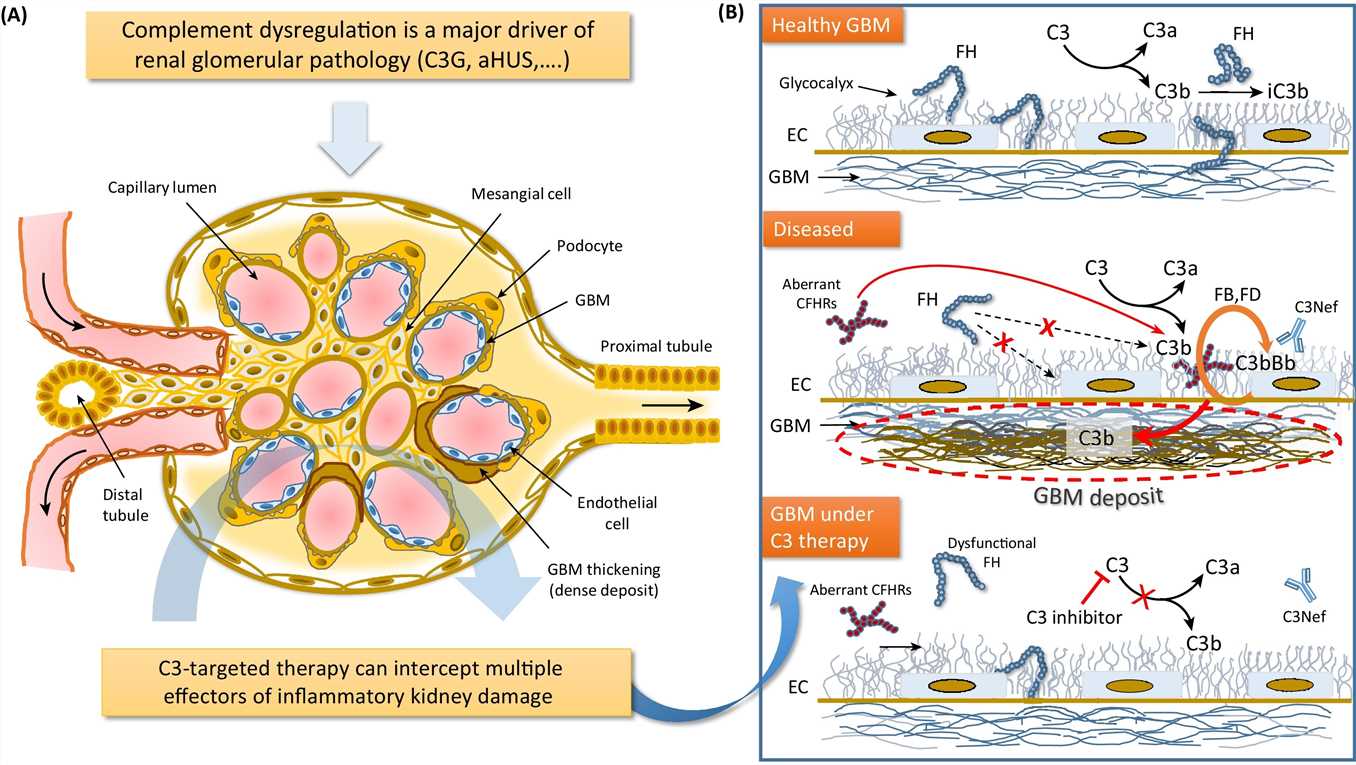
C3 glomerulotherapy.
The C3 glomerulopathies are a group of rare kidney diseases characterized by complement dysregulation occurring in the fluid phase and in the glomerular microenvironment, which results in prominent complement C3 deposition in kidney biopsy samples. The two major subgroups of C3 glomerulopathy — dense deposit disease (DDD) and C3 glomerulonephritis (C3GN) — have overlapping clinical and pathological features suggestive of a disease continuum. Dysregulation of the complement alternative pathway is fundamental to the manifestations of C3 glomerulopathy, although terminal pathway dysregulation is also common. Disease is driven by acquired factors in most patients — namely, autoantibodies that target the C3 or C5 convertases. These autoantibodies drive complement dysregulation by increasing the half-life of these vital but normally short-lived enzymes. Genetic variation in complement-related genes is a less frequent cause. No disease-specific treatments are available, although immunosuppressive agents and terminal complement pathway blockers are helpful in some patients. Unfortunately, no treatment is universally effective or curative. In aggregate, the limited data on renal transplantation point to a high risk of disease recurrence (both DDD and C3GN) in allograft recipients. Clinical trials are underway to test the efficacy of several first-generation drugs that target the alternative complement pathway.
What is MHRA in pharma?
The Medicines and Healthcare products Regulatory Agency (MHRA) is the government agency responsible for ensuring that medicines and medical devices work, and are acceptably safe. The MHRA keep watch over medicines and devices, and take any necessary action to protect the public promptly if there is a problem
What is NICE?
The National Institute for Health and Care Excellence (NICE) is an executive non-departmental public body of the Department of Health in England, which publishes guidelines in four areas:
- the use of health technologies within the National Health Service (England) and within NHS Wales (such as the use of new and existing medicines, treatments and procedures)
- clinical practice (guidance on the appropriate treatment and care of people with specific diseases and conditions)
- guidance for public sector workers on health promotion and ill-health avoidance
- guidance for social care services and users.
About Novartis:
Novartis is reimagining medicine to improve and extend people’s lives. As a leading global medicines company, they strive to use innovative science and digital technologies to create treatments in areas of great medical need. In our quest to find new medicines, they consistently rank among the world’s top companies investing in research and development. Novartis products reach more than 800 million people globally and they are finding innovative ways to expand access to our latest treatments. About 108,000 people of more than 140 nationalities work at Novartis around the world.
SOURCE:https://www.pharmatimes.com/news/novartis_uk_awarded_innovation_passport_for_investigational_therapy_1382697
For more Information: Sign-in Websites for Agrochemical & Pharmaceutical Databases:
Website: https://www.chemrobotics.com/ (Agrochemical Databases)
Website: https://chemroboticspharma.com/ (Pharmaceutical Databases)

