Synopsis :
- DUSA Pharmaceuticals Inc has reached settlement with Biofrontera to resolve a litigation over misappropriation of trade secrets and unfair practice, following which DUSA will receive USD 22.5 million (nearly Rs 170 crore).
- In 2018, Dusa Pharma had filed claims of patent infringement, trade secret misappropriation and wrongful interference against certain Biofrontera entities in the U.S. District Court for Massachusetts.
- The lawsuit also said Biofrontera had misappropriated confidential and trade secret information from Dusa and improperly obtained confidential information from former Dusa employees to sell and market Biofrontera’s own products.
On Tuesday said its arm DUSA Pharmaceuticals Inc has reached settlement with Biofrontera to resolve a litigation over misappropriation of trade secrets and unfair practice, following which DUSA will receive USD 22.5 million (nearly Rs 170 crore). In 2018, DUSA Pharmaceuticals had filed a suit at the US District Court of Massachusetts against Biofrontera Inc Biofrontera Bioscience GmbH, Biofrontera Pharma GmbH, and Biofrontera AG (together known as Biofrontera) alleging “misappropriation of trade secrets, tortious interference of contract and unfair trade practices”.
“We would like to update you that DUSA has reached a settlement with Bionfrontera resolving the litigation. Pursuant to the terms of the settlement, DUSA will receive USD 22.5 million from Biofrontera,” Sun Pharma said in a regulatory filing.

DUSA Pharmaceuticals Agree to Settlement with Biofrontera
The homegrown pharma major, however, did not disclose details of the settlement saying “additional details regarding the settlement are confidential”.
The company further said as required by law the parties have filed a notice of settlement with the US District Court and after 30 days if Biofrontera complies with the terms of the settlement agreement, the court will enter an order dismissing the litigation.
DUSA’s lawsuit against Biofrontera pertained to alleged trade secret misappropriation and patent infringement of its photodynamic therapy patents covering the company’s product, Levulan Kerastick (aminolevulinic acid HCl) for topical solution, used in the treatment of actinic keratoses — rough, scaly patch on the skin that develops from years of sun exposure. On Nov. 30, Sun Pharma said in an exchange filing that the companies have decided to settle the lawsuit. Accordingly, Dusa will receive a compensation of $22.5 million (Rs 168.9 crore).
The companies have filed the settlement notice with the District Court and the order for dismissal of litigation will be passed after 30 days, if Biofrontera complies with all terms of settlement.
Shares of Sun Pharma were trading 0.15% higher at 3:26 p.m. on Tuesday compared with the benchmark S&P BSE Sensex’s 0.55% fall
Aminolevulinic Acid HCl
Aminolevulinic acid is used in combination with photodynamic therapy (PDT; special blue light) to treat actinic keratoses (small crusty or scaly bumps
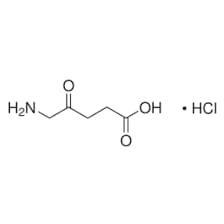
Aminolevulinic Acid Structure
Why is this medication prescribed?
Aminolevulinic acid is used in combination with photodynamic therapy (PDT; special blue light) to treat actinic keratoses (small crusty or scaly bumps or horns on or under the skin that result from exposure to sunlight and can develop into skin cancer) of the face or scalp. Aminolevulinic acid is in a class of medications called photosensitizing agents. When aminolevulinic acid is activated by light, it damages the cells of actinic keratosis lesions.
How should this medicine be used?
Aminolevulinic acid comes in a special applicator to be made into a solution and applied to the affected skin area by a doctor. You must return to the doctor 14 to 18 hours after aminolevulinic acid application to be treated by blue light PDT. For example, if you have Aminolevulinic acid applied in the late afternoon, you will need to have the blue light treatment the next morning. You will be given special goggles to protect your eyes during blue light treatment.
Do not put a dressing or bandage on the area treated with Aminolevulinic acid. Keep the treated area dry until you go back to the doctor for blue light treatment.
Your doctor will examine you 8 weeks after aminolevulinic acid and PDT treatment to decide whether you need retreatment of the same skin area.
What side effects can this medication cause?
- Tingling, stinging, prickling, or burning of lesions during blue light treatment (should get better within 24 hours)
- Redness, swelling, and scaling of treated actinic keratoses and surrounding skin (should get better within 4 weeks)
- Discoloration of the skin
- itching
- Bleeding
- Blistering
- Pus under the skin
- Hives
About Sun Pharma
Sun Pharmaceutical Industries Limited (d/b/a Sun Pharma) is an Indian multinational pharmaceutical company headquartered in Mumbai, Maharashtra, that manufactures and sells pharmaceutical formulations and active pharmaceutical ingredients (APIs) primarily in India and the United States. Sun Pharma has presence in more than 100 countries across the globe. It is largest pharma company in India and the fourth largest specialty generic pharmaceutical company in the world, with a total revenue of over US$4.5 billion as of June 2021. The products cater to a vast range of therapeutic segments covering psychiatry, anti-infectives, neurology, cardiology, orthopaedic, diabetology, gastroenterology, ophthalmology, nephrology, urology, dermatology, gynaecology, respiratory, oncology, dental and nutritionals.
Its API products include Acamprosate Calcium, Alendronate Sodium, Amifostine trihydrate, Budensonide and Carvedilol
DUSA Pharmaceuticals
DUSA Pharmaceuticals, Inc., owned by Sun Pharma, is a pharmaceutical company with dermatology products, including Levulan and Nicomide.
In the first quarter of 2006, DUSA Pharmaceuticals obtained Sirius Laboratories in a merger estimated to be worth 30 million dollars. Sirius was a privately held dermatology specialty pharmaceuticals company founded by Dr. Joel E. Bernstein in 2000 dedicated to the treatment of acne vulgaris and acne rosacea.
In 2012, Caraco Pharmaceutical Laboratories (CPL), a subsidiary of Sun Pharmaceutical, acquired DUSA
About Biofrontera AG
Biofrontera AG is a biopharmaceutical company specializing in the development and sale of dermatological drugs and medical cosmetics. The Germany-based company, with about 150 employees worldwide, develops and markets innovative products for the care, protection and treatment of the skin.


