Summary
Dr Reddy’s Laboratories, an Indian generic pharmaceutical company, has agreed to pay $61 million to Novartis AG for the cardiovascular brand Cidmus (approximately Rs 463 crore).
This comes weeks after Dr Reddy’s inking an exclusive sales and distribution agreement with the Switzerland-based global healthcare giant to buy the latter’s established medicines that included the Voveran range, the Calcium range, and Methergine.

Dr. Reddy’s will Pay Rs 463 Crore for Novartis’ Cardiovascular Drug Cidmus (Valsartan +Sacubitril)
Under the latest agreement, Dr Reddy’s will be assigned and transferred the Cidmus trademark in India from Novartis AG. Cidmus saw sales of Rs 136.4 crore in India for the most recent twelve months ending February 2022.
About Cidmus
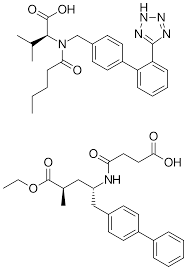
Structure of Cidmus (Valsartan +Sacubitril)
- Active Pharmaceutical Ingredient – Combination of two drug Sacubitril and Valsartan
- Medical Use – It belongs to a class of Cardiovascular Agents that is used in the Treatment of Heart Failure.
Mechanism of Action :
LCZ696 (Sacubitril/Valsartan) exhibits the novel mechanism of action of an angiotensin receptor neprilysin inhibitor (ARNI)
Valsartan blocks the angiotensin II receptor type 1 (AT1). This receptor is found on both vascular smooth muscle cells, and on the zona glomerulosa cells of the adrenal gland which are responsible for aldosterone secretion. In the absence of AT1 blockade, angiotensin causes both direct vasoconstriction and adrenal aldosterone secretion, the aldosterone then acting on the distal tubular cells of the kidney to promote sodium reabsorption which expands extracellular fluid (ECF) volume. Blockade of (AT1) thus causes blood vessel dilation and reduction of ECF volume.
Sacubitril is a prodrug that is activated to sacubitrilat (LBQ657) by de-ethylation via esterases. Sacubitrilat inhibits the enzyme neprilysin, a neutral endopeptidase that degrades vasoactive peptides, including natriuretic peptides, bradykinin, and adrenomedullin. Thus, sacubitril increases the levels of these peptides, causing blood vessel dilation and reduction of ECF volume via sodium excretion.
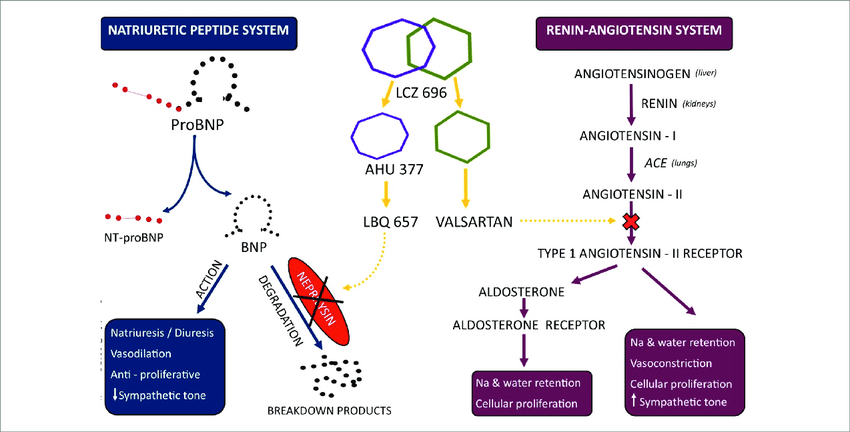
LCz696 mechanism of action
In a statement on April 1, Dr Reddy’s said the Cidmus brand shall be affixed on the pharmaceutical composition comprising a combination of Valsartan and Sacubitril (currently under Novartis patent) which is indicated for heart failure patients with reduced ejection fraction.
The Hyderabad headquartered drugmaker said it will look to leverage its wide base to engage with healthcare professionals and to significantly enhance the reach of the product in and beyond metros into tier-I and tier-II markets in India through its strong marketing and distribution network to maximise access to patients in India.
Dr Reddy’s said given the prevalence of cardiovascular diseases, this acquisition will allow it to make a trusted portfolio of medicines available to patients in India and that Cidmus will be a strong addition to its existing portfolio in the cardiovascular segment alongside its leading brands such as Stamlo, Stamlo Beta, Reclide-XR and Reclimet-XR.

