Summary :
- The U.S. Food and Drug Administration approved Olumiant (baricitinib) oral tablets to treat adult patients with severe alopecia areata, a disorder that often appears as patchy baldness and affects more than 300,000 people in the U.S. each year.
- Today’s action marks the first FDA approval of a systemic treatment (i.e. treats the entire body rather than a specific location) for alopecia areata.
“Access to safe and effective treatment options is crucial for the significant number of Americans affected by severe alopecia,” said Kendall Marcus, M.D., director of the Division of Dermatology and Dentistry in the FDA’s Center for Drug Evaluation and Research. “Today’s approval will help fulfill a significant unmet need for patients with severe alopecia areata.”
About Olumiant (Baricitinib)
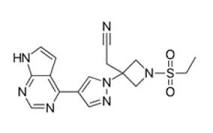
Baricitinib Structure
API – Baricitinib
Description – Baricitinib, sold under the brand name Olumiant among others, is a medication used for the treatment of rheumatoid arthritis, alopecia areata, and COVID-19. It acts as an inhibitor of janus kinase, blocking the subtypes JAK1 and JAK2.
Indication : In the US and Europe, baricitinib is indicated for the treatment of adult patients with moderately to severely active rheumatoid arthritis who have had an inadequate response to one or more TNF blockers. Baricitinib may be used as monotherapy or in combination with methotrraxate or other DMARDs.
- In Europe, baricitinib is indicated for the treatment of moderate to severe atopic dermatitis in adult patients who are candidates for systemic therapy.
- In the US, baricitinib is also indicated for the treatment of coronavirus disease 2019 (COVID-19) in hospitalized adults requiring supplemental oxygen, non-invasive or invasive mechanical ventilation, or extracorporeal membrane oxygenation.9 Recently, it is also approved as the treatment for severe alopecia areata in adults
Mechanism of action
As members of the tyrosine kinase family, Janus kinases (JAKs) are intracellular enzymes that modulate signals from cytokines and growth factor receptors involved in hematopoiesis, inflammation, and immune cell function. Upon binding of extracellular cytokines and growth factors, JAKs phosphorylate and activate Signal Transducers and Activators of Transcription (STATs). STATs modulate intracellular activity, including gene transcription of inflammatory mediators that promote an autoimmune response, such as IL-2, IL-6, IL-12, IL-15, IL-23, IFN-γ, GM-CSF, and interferons. The JAK-STAT pathway has been implicated in the pathophysiology of rheumatoid arthritis, as it is associated with an overproduction of inflammatory mediators.
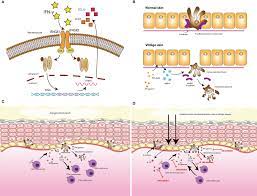
Baricitinib Moa in Alopecia areata
There are four JAK proteins: JAK 1, JAK 2, JAK 3 and TYK2. JAKs form homodimers or heterodimers and pair differently in different cell receptors to transmit cytokine signaling. Baricitinib is a selective and reversible inhibitor of JAK1 and JAK2 with less affinity for JAK3 and TYK2; however, the relevance of inhibition of specific JAK enzymes to therapeutic effectiveness is not currently known. Baricitinib inhibits the activity of JAK proteins and modulates the signaling pathway of various interleukins, interferons, and growth factors. It was also shown to decrease the proliferation of JAK1/JAK2 expression in mutated cells and induce cell apoptosis.
Protein binding -Baricitinib is approximately 50% bound to plasma proteins and 45% bound to serum proteins.
Half-life -The elimination half-life in patients with rheumatoid arthritis is approximately 12 hours. The elimination half-life was 10.8 hours in intubated patients with COVID-19 who received baricitinib via nasogastric (NG) or orogastric (OG) tube.
Olumiant was originally approved in 2018. It is approved as a treatment for certain adult patients with moderately to severely active rheumatoid arthritis. Olumiant is also approved for the treatment of COVID-19 in certain hospitalized adults.
The FDA granted the approval of Olumiant to Eli Lilly and Company.
About Alopecia Areata
Alopecia areata, commonly referred to as just alopecia, is an autoimmune disorder in which the body attacks its own hair follicles, causing hair to fall out, often in clumps. Olumiant is a Janus kinase (JAK) inhibitor which blocks the activity of one or more of a specific family of enzymes, interfering with the pathway that leads to inflammation.
- Alopecia areata occurs when the immune system attacks hair follicles and may be brought on by severe stress.
- The main symptom is hair loss.
- Treatment may address any underlying conditions and includes topical scalp medication.
About Eli Lilly and Company
Eli Lilly and Company is an American pharmaceutical company headquartered in Indianapolis, Indiana, with offices in 18 countries. Its products are sold in approximately 125 countries. The company was founded in 1876 by, and named after, Colonel Eli Lilly, a pharmaceutical chemist and veteran of the American Civil War.
Lilly was founded in 1876 by Colonel Eli Lilly, a man committed to creating high-quality medicines that met real needs in an era of unreliable elixirs peddled by questionable characters.
Weblink: https://www.chemrobotics.com
- AgroPat Lite– Access 5500 pesticides with chemistry, Biology, Regulatory, and IP info. Covers the product information including formulation, combination, developer, innovator, existing intellectual property, regulatory requirement, biology data including spectrum, MOA, DFU, toxicity profile, and safety. (Designed for Business Development function)
-
-
- AgroPat Ultimate– In detailed Access 5500 pesticides with chemistry, Biology, Regulatory, and IP info. (Designed for Research & Development function)
- Indian Medicine Database –Approved Drugs, Medical Devices, Approved Regenerative Medical Products
- Weblink: https://imd.chemrobotics.com/
- Indian Pesticide Database (IPD)– All Indian Approvals, e.g. 9(3) and 9(4), etc.
- Global Agro Product Directory(More than 55countries approved product info. with relevant documents such as label, factsheet and monograph)
- Weblink: https://www.chemrobotics.com/pesticides-directory/
- Global MRL Database(More than 85 countries MRL info.)
- Jarvis– A Competitor Patents Watch Database for Agrochemical
- Technical Routes(More than 15000 routes of synthesis for Agrochemical & Pharmaceutical)
- Technical Suppliers(Provides technical supplier information)
- Company Directory– KSM Supplier(s) Database — More than 10 K Companies listed from Pharma / Agrochemical / Fine Chemical Domain with their product offering in Pharma / Agrochemical / Fine Chemical segment,
- Weblink: https://companydirectory.chemrobotics.com
- ChemRobotics SPC Database– Provides Patent SPC data Europe
- PharmVetPat –Access chemistry including ROS, KSM, Intermediate, Biology, Regulatory, and IP info for all pharm molecules.
-


