Summary –
- New IND Expands Liver Disease Pipeline for CRV431
- IND Clearance Allows CRV431 to Move Directly into Phase 2 for Hepatocellular Carcinoma Treatment
- New IND Provides for Complementary Therapeutic Approach of Treating NASH and Liver Cancer with a Single Once-a-Day Oral Agent
Hepion Pharmaceuticals, Inc. (NASDAQ:HEPA), a clinical stage biopharmaceutical company focused on Artificial Intelligence (“AI”)-driven therapeutic drug development for the treatment of non-alcoholic steatohepatitis (“NASH”) and other liver diseases, today announced that the U.S. Food and Drug Administration (“FDA”) has accepted its investigational new drug (“IND”) application for CRV431, a liver-targeting, novel cyclophilin inhibitor, for the treatment of hepatocellular carcinoma (“HCC”).
On July 29, 2019, Hepion received FDA authorization of an IND to initiate the study of CRV431 for the treatment of NASH. To date, Hepion has completed Phase 1 studies in healthy volunteers and, more recently, announced positive data from its Phase 2a ‘AMBITION’ trial in subjects with presumed F2 and F3, where CRV431 was well tolerated, and all primary endpoints were met. A larger Phase 2b NASH study of over 300 subjects with paired liver biopsies, called ‘ASCEND-NASH’, is expected to be initiated in 2022. ASCEND-NASH will evaluate CRV431’s effects on the histologic endpoints of liver steatosis and fibrosis over 12 months dosing.
“Liver cancer is the sixth most prevalent cancer worldwide and is the second most common cause of cancer death,”1 commented Todd Hobbs, MD, Hepion’s Chief Medical Officer. “The most common form of liver cancer is HCC, which comprises about 90% of all liver cancers. Major risk factors associated with development of HCC include NASH, liver fibrosis and cirrhosis, viral hepatitis, chronic alcohol consumption, and metabolic syndrome. As the global prevalence of NASH is increasing, the incidence of HCC arising from NASH is also increasing. Approximately 25% to 30% of NASH-related HCC develops in the absence of cirrhosis and, therefore, occurs without many of the symptomatic warning signs of this aggressive form of cancer. An orally administered drug that simultaneously targets NASH and HCC would offer an advantageous therapeutic strategy to patients suffering these potentially life-threatening conditions. CRV431’s novel potential stems from its pleiotropic pharmacologic activities and from its ability to target the liver, allowing for a drug candidate ideally suited for the treatment of liver disease.”

FDA IND Application Clearance to Hepion Pharma for its CRV431 in the Treatment of Liver Cancer
Dr. Hobbs continued, “Another potentially important benefit is the patient experience while taking CRV431. Most cancer drugs are delivered by injection and are associated with significant side effects. In contrast, CRV431 is orally administered and has been shown to be well tolerated in clinical trials to date. We are optimistic that CRV431 may provide significant anti-cancer effects without imposing additional challenges and distress often associated with cancer drugs.”
Robert Foster, PharmD, PhD, Hepion’s CEO, said, “Increased cyclophilin isoform expression has been associated with negative outcomes in HCC. Importantly, CRV431 potently inhibits many of these isoforms in humans. Cyclophilins are enzymes that regulate many molecular and cellular activities that become dysregulated both in NASH and HCC. These dysregulated activities can lead to aberrations in signal transduction pathways, cell proliferation, cell death, extracellular environment including increased fibrosis, energy metabolism, inflammation, and immunity. Therapeutic intervention with CRV431 administration may reduce the pathologic potential associated with heightened cyclophilin activities in NASH and HCC, potentially allowing for a return to a healthier state.”
Dr. Foster continued, “CRV431 has shown anti-tumor activity in multiple animal studies, and our research team has been intensively investigating the specific mechanisms that have produced these outcomes. Our investigations have revealed interesting direct and indirect effects of CRV431 on cancer cells and tumors. For example, gene expression analyses have demonstrated CRV431’s ability to attenuate drug-resistance pathways and Wnt-β-catenin-Myc signaling, the latter being mutationally over-activated in 30% to 50% of human HCC tumors. We recently also found that CRV431 increased lymphocyte infiltration into liver tumors in a manner similar to an immune checkpoint inhibitor, also known as an anti-PD-1 antibody, which is considered to be an important anti-tumor approach. We believe that the wide array of pharmacologic activities offered by CRV431 in the treatment of NASH and HCC should bode well for the further clinical development in both important indications.”
About CRV431
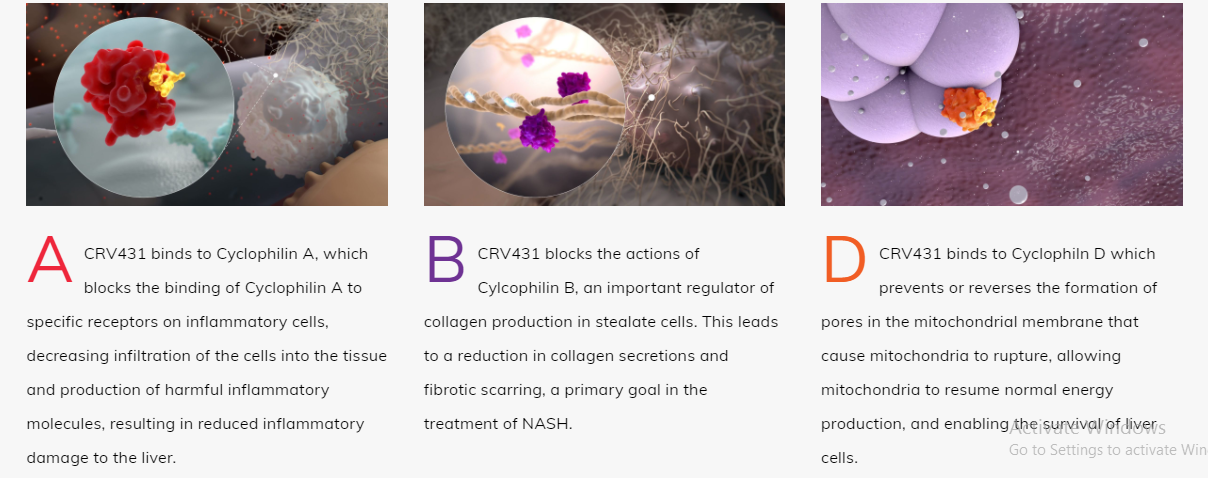
CRV431 is Hepion’s clinical phase, lead oral drug candidate for nonalcoholic steatohepatitis (NASH) and viral hepatitis-induced liver disease. CRV431 meets all of Hepion’s criteria for an effective and unique pleiotropic liver disease drug. As a novel molecule chemically derived from a pharmacological class of therapeutic drugs used clinically for over 35 years, CRV431 has thus far demonstrated an excellent safety profile in all animal and clinical studies to date. Thanks to multiple, diverse mechanisms of action and its accumulation in the liver (5-times higher concentrations than in the blood), CRV431 has demonstrated efficacy in two distinct animal models of NASH and HBV replication. These findings are consistent with in vitro studies documenting mechanisms of action as an antiviral agent (HBV, HCV, HDV ), protection from cellular stress and death, anti-steatotic, anti-inflammatory, anti-fibrotic, and anti-cancer activities. Thus, CRV431 is positioned to become a highly versatile therapeutic drug for the most prominent liver diseases of our time. No other marketed drugs or NASH drug candidates in development address all these mechanisms.
CANDIDATE CRV431 MECHANISMS OF ACTION IN CHRONIC LIVER DISEASE (NASH AND VIRAL HEPATITIS)
CRV431 specifically binds cyclophilin isomerase enzymes and inhibits cyclophilin function with the highest known potency of any reported cyclophilin inhibitor (Ki= 1 nM). The pleiotropic actions of CRV431 are due to the fact that multiple cyclophilin isoforms exist in the body and participate in many biological processes. Cyclophilins assume especially prominent roles in disease processes such as cell death, fibrosis, and cancer cell growth and metastasis. Many viruses have evolved to recruit cyclophilins into their life cycles to assist in viral replication and evade the immune system. By blocking the participation of cyclophilins in these processes, CRV431 displays a variety of therapeutic activities.
About Hepion Pharmaceuticals
The Company’s lead drug candidate, CRV431, is a potent inhibitor of cyclophilins, which are involved in many disease processes. CRV431 is currently in clinical-phase development for the treatment of NASH, with the potential to play an important role in the overall treatment of liver disease – from triggering events through to end-stage disease. CRV431 has been shown to reduce liver fibrosis and hepatocellular carcinoma tumor burden in experimental models of NASH; and has demonstrated antiviral activities towards HBV, HCV, and HDV through several mechanisms, in nonclinical studies.
Hepion has created a proprietary AI platform, called AI-POWR™, which stands for Artificial Intelligence – Precision Medicine; Omics (including genomics, proteomics, metabolomics, transcriptomics, and lipidomics); World database access; and Response and clinical outcomes. Hepion intends to use AI-POWR™ to help identify which NASH patients will best respond to CRV431, potentially shortening development timelines and increasing the delta between placebo and treatment groups. In addition to using AI-POWR™ to drive its ongoing NASH clinical development program, Hepion intends to use the platform to identify additional potential indications for CRV431 to expand the company’s footprint in the cyclophilin inhibition therapeutic space.

