Summary-The Food and Drug Administration granted accelerated approval to teclistamab-cqyv (Tecvayli, Janssen Biotech, Inc.), the first bispecific B-cell maturation antigen (BCMA)-directed CD3 T-cell engager, for adult patients with relapsed or refractory multiple myeloma who have received at least four prior lines of therapy, including a proteasome inhibitor, an immunomodulatory agent, and an anti-CD38 monoclonal antibody.

johnson and johnson
The Janssen Pharmaceutical Companies of Johnson & Johnson announced today that the U.S. Food and Drug Administration (FDA) approved TECVAYLI™ (teclistamab-cqyv) for the treatment of adult patients with relapsed or refractory multiple myeloma, who previously received four or more prior lines of therapy, including a proteasome inhibitor, immunomodulatory drug and anti-CD38 monoclonal antibody. TECVAYLI™ is a first-in-class, bispecific T-cell engager antibody that is administered as a subcutaneous treatment. This off-the-shelf (or ready to use) therapy uses innovative science to activate the immune system by binding to the CD3 receptor expressed on the surface of T-cells and to the B-cell maturation antigen (BCMA) expressed on the surface of multiple myeloma cells and some healthy B-lineage cells.
This indication is approved under accelerated approval based on response rate. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trial(s).
About TECVAYLI
- API– teclistamab-cqyv
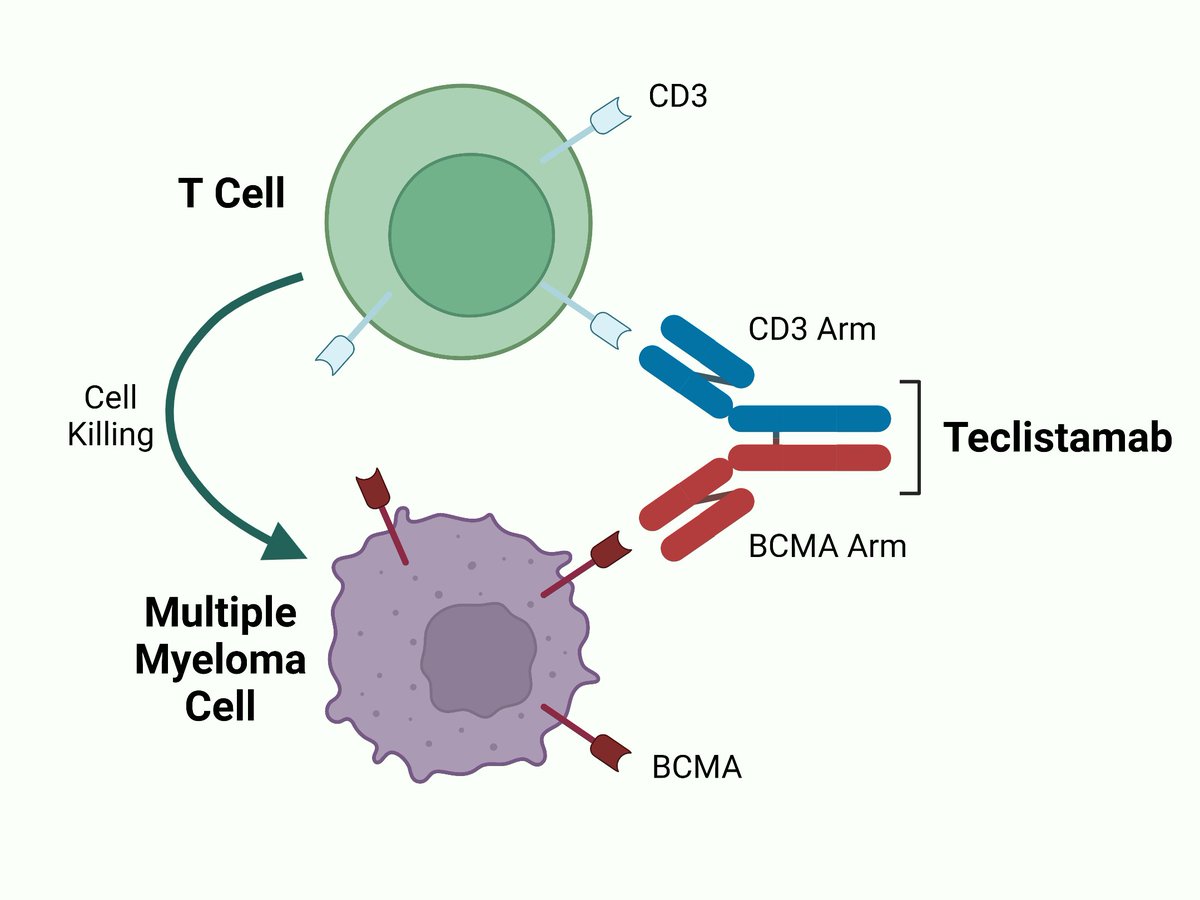
Teclistamab Structure
- Description-Teclistamab is an IgG4-PAA bispecific antibody that targets the CD3 receptor expressed on the surface of T cells and B cell maturation antigen (BCMA) expressed on malignant multiple myeloma cells. Teclistamab consists of an anti-BCMA arm and an anti-CD3 arm connected via two interchain disulfide bonds, allowing the drug to recruit CD3-expressing T cells to BCMA-expressing cells to promote T cell–mediated cytotoxicity. On August 24, 2022, the European Commission (EC) granted conditional marketing authorization of teclistamab as first-in-class bispecific antibody for the treatment of multiple myeloma, marking its first global approval.Teclistamab was later granted accelerated approval by the FDA on October 25, 2022 .TECVAYLI™ is Janssen’s fourth approved treatment for multiple myeloma, further diversifying the company’s industry-leading oncology portfolio and deepening its commitment to discovering and developing therapies for this rare blood cancer.
- Biologic Classification -Protein Based Therapies (Monoclonal antibody (mAb)
- Indication-Teclistamab is indicated as monotherapy for the treatment of adult patients with relapsed and refractory multiple myeloma who have received at least three prior therapies, including an immunomodulatory agent, a proteasome inhibitor, and an anti-CD38 antibody and have demonstrated disease progression on the last therapy. Teclistamab is approved by the EC and FDA under conditional marketing authorization and accelerated approval, respectively. New evidence for this drug will be continuously monitored and reviewed, which will affect continued approval for the drug’s indication.
- Class-Antineoplastics; Bispecific antibodies; Immunotherapies
- Mechanism of Action-Antibody-dependent cell cytotoxicity; Cytotoxic T lymphocyte stimulants
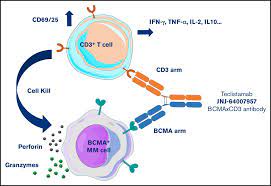
Teclistamab-cqyv moa
- Orphan Drug Status-Yes – Multiple myeloma
- Volume of distribution-The mean (CV%) volume of distribution of teclistamab was 5.63 L (29%). The volume of distribution of teclistamab increases with increasing body weight.
- Half-life-Based on non-compartmental analysis, the mean half-life (SD) was 3.8 (1.7) days (individual values ranging up to 8.8 days) following the first treatment intravenous dose of teclistamab.
The pivotal Phase 2 MajesTEC-1 clinical trial included patients who had received a median of five prior lines of therapy (n=110). An overall response rate (ORR) of 61.8 percent (95 percent Confidence Interval [CI]: 52.1 percent, 70.9 percent) was achieved, notably with 28.2 percent of patients achieving a complete response (CR) or better (CR or stringent complete response [sCR]). The median time to first response was 1.2 months (range 0.2 to 5.5 months). With a median follow-up of 7.4 months, the estimated duration of response (DOR) rate was 90.6 percent (95 percent CI: 80.3 percent, 95.7 percent) at six months and 66.5 percent (95 percent CI: 38.8 percent, 83.9 percent) at nine months. The study included heavily pretreated patients, and 78 percent of patients received four or more prior lines of therapy. All patients were triple-class exposed (to a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody), and 76 percent were triple-class refractory (to a proteasome inhibitor, an immunomodulatory agent and an anti-CD38 monoclonal antibody).
“Today’s achievement, which marks an important addition to our diverse and growing oncology portfolio, strengthens our resolve to discover and develop much-needed cancer treatments for patients and physicians,” said Peter Lebowitz, M.D., Ph.D., Global Therapeutic Area Head, Oncology, Janssen Research & Development, LLC. “The approval of TECVAYLI, which demonstrated an overall response rate of more than 60 percent in heavily pretreated patients, underscores our commitment to translate science into medicines as we strive toward our goal of one day eliminating this disease.”
The Safety Information for TECVAYLI™ includes a boxed warning for Cytokine Release Syndrome (CRS) and Neurologic Toxicity including immune effector cell-associated neurotoxicity syndrome in addition to warnings and precautions for hepatotoxicity, infections, neutropenia, hypersensitivity and other administrative reactions and embryo-fetal toxicity. The most common adverse reactions (>20%) in the safety population of MajesTEC-1 (n=165) were pyrexia, CRS, musculoskeletal pain, injection site reaction, fatigue, upper respiratory tract infection, nausea, headache, pneumonia, and diarrhea. The most common Grade 3 to 4 laboratory abnormalities (>20%) were decreased lymphocytes, decreased neutrophils, decreased white blood cells, decreased hemoglobin and decreased platelets. TECVAYLI™ is available only through a restricted program called the TECVAYLI™ Risk Evaluation and Mitigation Strategy. Details of the Important Safety Information are included below. TECVAYLI™ is supplied as 30mg/3mL and 153mg/1.7mL single-dose vials.
About Multiple Myeloma
Multiple myeloma is an incurable blood cancer that affects a type of white blood cell called plasma cells, which are found in the bone marrow. In multiple myeloma, these plasma cells change, spread rapidly and replace normal cells in the bone marrow with tumors. In 2022, it is estimated that more than 34,000 people will be diagnosed with multiple myeloma, and more than 12,000 people will die from the disease in the U.S. While some people diagnosed with multiple myeloma initially have no symptoms, most patients are diagnosed due to symptoms that can include bone fracture or pain, low red blood cell counts, tiredness, high calcium levels, kidney problems or infections.
About the Janssen Pharmaceutical Companies of Johnson & Johnson
At Janssen, we’re creating a future where disease is a thing of the past. We’re the Pharmaceutical Companies of Johnson & Johnson, working tirelessly to make that future a reality for patients everywhere by fighting sickness with science, improving access with ingenuity, and healing hopelessness with heart. We focus on areas of medicine where we can make the biggest difference: Cardiovascular, Metabolism, & Retina; Immunology; Infectious Diseases & Vaccines; Neuroscience; Oncology; and Pulmonary Hypertension.
Weblink: https://www.chemrobotics.com
- AgroPat Lite– Access 5500 pesticides with chemistry, Biology, Regulatory, and IP info. Covers the product information including formulation, combination, developer, innovator, existing intellectual property, regulatory requirement, biology data including spectrum, MOA, DFU, toxicity profile, and safety. (Designed for Business Development function)
-
- AgroPat Ultimate– In detailed Access 5500 pesticides with chemistry, Biology, Regulatory, and IP info. (Designed for Research & Development function)
- Indian Medicine Database –Approved Drugs, Medical Devices, Approved Regenerative Medical Products
- Weblink: https://imd.chemrobotics.com/
- Indian Pesticide Database (IPD)– All Indian Approvals, e.g. 9(3) and 9(4), etc.
- Global Agro Product Directory(More than 55countries approved product info. with relevant documents such as label, factsheet and monograph)
- Weblink: https://www.chemrobotics.com/pesticides-directory/
- Global MRL Database(More than 85 countries MRL info.)
- Jarvis– A Competitor Patents Watch Database for Agrochemical
- Technical Routes(More than 15000 routes of synthesis for Agrochemical & Pharmaceutical)
- Technical Suppliers(Provides technical supplier information)
- Company Directory– KSM Supplier(s) Database — More than 10 K Companies listed from Pharma / Agrochemical / Fine Chemical Domain with their product offering in Pharma / Agrochemical / Fine Chemical segment,
- Weblink: https://companydirectory.chemrobotics.com
- ChemRobotics SPC Database– Provides Patent SPC data Europe
- PharmVetPat –Access chemistry including ROS, KSM, Intermediate, Biology, Regulatory, and IP info for all pharm molecules.
-
- Weblink: https://chemroboticspharma.com/pharmVetPat

