Summary :
- Qelbree (i.e. SPN-812) is a nonstimulant ADHD medication containing a serotonin norepinephrine modulating agent approved by the FDA to treat attention deficit hyperactivity disorder (ADHD or ADD) in patients 6 to 17 years of age.
- First novel, nonstimulant option for adults with ADHD in 20 years
- ADHD affects an estimated 10 million adults in the U.S.
Supernus Pharmaceuticals, Inc. (Nasdaq: SUPN), a biopharmaceutical company focused on developing and commercializing products for the treatment of central nervous system (CNS) diseases, announced today that the U.S. Food and Drug Administration (FDA) approved an expanded indication for Qelbree (viloxazine extended-release capsules) for the treatment of attention deficit hyperactivity disorder (ADHD) in adult patients aged 18 and older. The FDA has now approved Qelbree for the treatment of ADHD in children (starting at age 6), adolescents and adults.
“Until today, nonstimulant ADHD options for adults have been very limited,” said Greg Mattingly, M.D, founding partner of St. Charles Psychiatric Associates in St. Louis, Mo. “This approval is positive news and offers a new novel option for the millions of American adults who are trying to find the right treatment to manage their ADHD symptoms.”
About Qelbree®
Qelbree is a prescription medicine used to treat attention deficit hyperactivity disorder (ADHD) in adults and children 6 years of age and older. It is not known if this medicine is safe and effective in children less than 6 years of age.
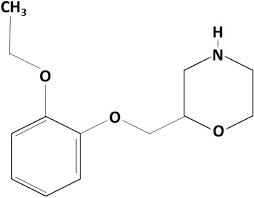
Viloxazine Structure
- API-:Viloxazine
- Inactive Ingredients: ammonium hydroxide, black iron oxide, butyl alcohol, corn starch, ethylcellulose, FD&C Blue #1, FD&C Red #28, FD&C Yellow #5, FD&C Yellow #6, FD&C Yellow #10, gelatin, hypromellose, isopropyl alcohol, lactose monohydrate, medium chain triglycerides, oleic acid, polyethylene glycol, potassium hydroxide, propylene glycol, shellac, strong ammonia solution, sucrose, talc, triacetin, titanium dioxide.
- Indication : Viloxazine is indicated to treat attention deficit hyperactivity disorder (ADHD) in people aged six through seventeen.
Mechanism of Action :
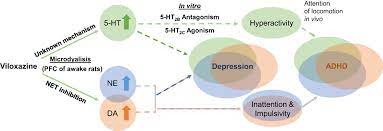
Viloxazine MOA
Qelbree™ (viloxazine extended-release capsules), is now available for the treatment of attention-deficit hyperactivity disorder (ADHD) in patients 6 to 17 years of age. While the mechanism of action of Qelbree in the treatment of ADHD is unclear, it is thought to be through inhibiting the reuptake of norepinephrine
Side Effect
The most common side effects in children 6 to 17 years of age include:
- sleepiness
- tiredness
- vomiting
- irritability
- decreased appetite
- nausea
- trouble sleeping
The most common side effects in adults include:
- insomnia
- sleepiness
- nausea
- dry mouth
- headache
- tiredness
- decreased appetite
- constipation
Qelbree is a novel nonstimulant taken once-daily for full-day exposure. Efficacy and symptom improvement was observed early in treatment. It has a proven safety and tolerability profile, with no evidence of abuse potential in clinical studies. The approval is based on positive results from a randomized, double blind, placebo-controlled Phase III study of Qelbree in adults with ADHD and represents the first approval of a novel nonstimulant treatment for adults in 20 years.
“As a leader in the field of CNS, we are fully committed to better understanding how to treat complex diseases such as ADHD,” said Jack Khattar, President and CEO of Supernus Pharmaceuticals. “Today’s approval marks a major advancement in the treatment of ADHD and is an important milestone just one year after the approval of Qelbree to treat pediatric patients. We are proud to bring a new novel nonstimulant option for adults into the market after two decades.”
About Supernus Pharmaceuticals, Inc.
Supernus Pharmaceuticals is a biopharmaceutical company focused on developing and commercializing products for the treatment of central nervous system (CNS) diseases.
Our diverse neuroscience portfolio includes approved treatments for epilepsy, migraine, ADHD, hypomobility in Parkinson’s disease (PD), cervical dystonia, chronic sialorrhea, dyskinesia in PD patients receiving levodopa-based therapy, and drug-induced extrapyramidal reactions in adult patients. We are developing a broad range of novel CNS product candidates including new potential treatments for hypomobility in PD, epilepsy, depression, and other CNS disorders.
For more Information: Sign in Websites for Agrochemical & Pharmaceutical Databases:
Website : https://www.chemrobotics.com/ (Agrochemical Databases)
Website : https://chemroboticspharma.com/ (Pharmaceutical Databases)


